1)
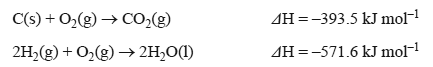
Given the information above, what is the enthalpy change for the following reaction?
C(s) + 2H2O(l) => CO2(g) + 2H2(g)
A. –965.1 kJ mol–1
B. –107.7 kJ mol–1
C. +178.1 kJ mol–1
D. +679.3 kJ mol–1
Solution

2) What is the enthalpy change when 40 g of NaOH is dissolved in one litre of water, given that the temperature of the
solution increased by 10.6 °C?
A. – 0.44 kJ mol–1
B. – 4.4 kJ mol–1
C. – 44 kJ mol–1
D. – 440 kJ mol–1
Solution
3) Sketch the energy profile for the complete combustion of a methyl ester using the axis below, labelling
the energy of the reactants, the products and the activation energy.
Solution

4) Olive oil, which has been part of the human diet for thousands of years, is derived from the fruit of the olive tree. 1.00 g of olive oil is burned in a bomb calorimeter with excess pure oxygen.
The calibration factor of the calorimeter is 9112 J °C–1. The burning of the olive oil increased the temperature in the bomb calorimeter from 20.0 °C to 22.4 °C.
a) Calculate the heat released by 1.00 g of olive oil.
Solution

b) Assuming the only constituent of olive oil is glycerol trioleate(C57H104O6.), write a combustion reaction for this molecule.
Solution
