1) Biodiesel is a mixture of fatty acid methyl esters. A particular triglyceride used in the manufacture of biodiesel was analysed by reacting it with excess methanol and a potassium hydroxide catalyst. This reaction produced fatty acid methyl esters and glycerol. At the conclusion of the reaction, two liquid layers were observed in the reaction vessel. The bottom layer was an aqueous solution.
a. Other than water, name one substance that would be found in the aqueous layer. Justify your answer.
Solution

b) The top layer is a non-aqueous mixture. It was separated from the aqueous layer and then purified. The non-aqueous layer was found to contain the fatty acid methyl esters. A small sample of the purifi ed ester mixture was passed through a gas chromatograph (GC) attached to a mass spectrometer. The chromatogram showed two peaks, indicating that the ester mixture contained two different fatty acid
methyl esters, A and B. The peak area of each compound and the mass-to-charge ratio of the molecular ion of each compound are shown in the following table. Assume that the charge on each molecular ion is +1.

The mass spectrum of methyl ester A corresponds to that of methyl palmitate, CH3(CH2)14COOCH3. What are the name and semi-structural formula of methyl ester B? (Refer to 'Formulas of some fatty acids' in the data book.)
Solution

c) Use the information above to draw a structure of the triglyceride. Use semi-structural formulas to represent the fatty acid residues.
Solution

2) Maltose is a sugar often used in the production of beer. The structure of maltose is shown below.
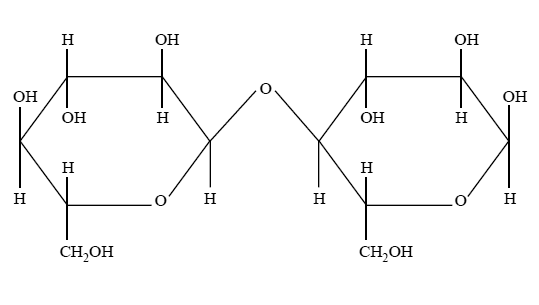
a) Draw a structure of the monomer from which maltose is derived.
Solution

b) Name of the monomer drawn in part a, identify the type of reaction that occurs when these monomers combine to form maltose and name the linkage joining the monomers in maltose.
Solution

3) Which one of the following is not true of protein denaturation?
A. It could result from a temperature change.
B. It may be caused by a pH change.
C. It alters the primary structure.
D. It results in a change in the shape of the protein.
Solution

4) Which compound of 24 carbon atoms has the least number of carbon–hydrogen, C–H, bonds?
A. a polypeptide that consists of four isoleucine residues
B. a molecule of lignoceric acid, which is a saturated fatty acid
C. a segment of polyethene that consists of 12 ethene residues
D. a molecule of maltotetraose, which is a polysaccharide that has four glucose residues

5) After a murder had been committed, a forensic chemist obtained crime scene blood samples and immediately placed them in two sterile containers labelled Sample I and Sample II.
a) The chemist discovered that Sample I contained a particular protein, which was analysed to reveal thefollowing sequence of amino acid residues.
-ser-gly-tyri.
Referring to the data book, draw the structure of this sequence of amino acid residues and circle one amide link/peptide bond in your drawing.
Solution

b) The protein was hydrolysed in the presence of a suitable enzyme and the amino acid glycine was isolated. The glycine sample was then dissolved in a 0.1 M solution of sodium hydroxide. Draw the structure of glycine in this solution.
Solution

c) Sample II was carefully treated to replicate and extract sections of the DNA. It was found that the DNA matched that of one of the murder suspects.
A section of the suspect's DNA contained a unique fault within the base sequence, -CAGCAG-, repeated many times.
What would be the base sequence matching this in the complementary strand?
Solution

d) What kind of bonding operates between base pairs? Is this bonding stronger or weaker than the bonding between the components in a single strand of DNA?
Solution

e) To what component of a DNA strand are the bases attached? What kind of bonding operates between this component and the base?
Solution
