1) A car manufacturer is planning to sell hybrid cars powered by a type of hydrogen fuel cell connected to a nickel metal hydride, NiMH, battery.
A representation of the hydrogen fuel cell is given below.
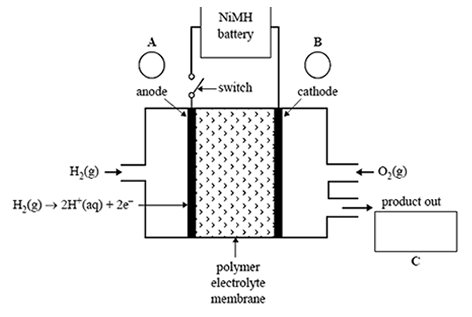
a)The overall cell reaction is
2H2(g) + O2(g) => 2H2O(g)
On the diagram above, indicate the polarity of the anode and the cathode in circles A and B, and identify the product of the reaction in box C
Solution

b) Write an equation for the reaction that occurs at the cathode when the switch is closed.
Solution

c) Identify one advantage and one disadvantage of using this fuel cell instead of a petrol engine to
power the car.
Solution
