1) The lithium-ion battery is a secondary cell that is now widely used in portable electronic devices. In these batteries, lithium ions, Li+, move through a special non-aqueous electrolyte between the two electrodes. The batteries are housed in sealed containers to ensure that no moisture can enter them. Both electrodes are made up of materials that allow the lithium ions to move into and out of their structures. The anode consists of LiC6, where lithium is embedded in the graphite structure. Lithium cobalt oxide, LiCoO2, is commonly used as the material in the cathode. The reaction at the cathode is quite complex. When the cell discharges, Li+ ions move out of the anode and enter the cathode. During discharge, the half-cell reaction at the anode is
LiC6 → Li+ + e + C6
a. During discharge, what is the polarity of the graphite electrode?
Solution
b. Write the half-equation for the reaction that occurs at the cathode of a lithium-ion battery when it is recharged.
Solution
c. In a lithium-ion battery, lithium metal must not be in contact with water. Explain why and justify your answer with the use of appropriate equations
Solution
d. Identify one design feature of the lithium-ion battery that enables it to be recharged.
Solution
e. What is one advantage of using a secondary cell compared to using a fuel cell?
Solution

2) The following diagram shows a galvanic cell that is set up in a university laboratory.
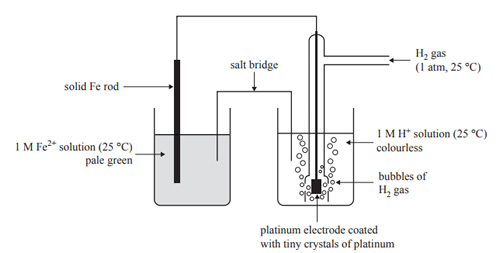
The half-cell on the right is called the standard hydrogen electrode (SHE). It is the standard against which all standard redox potentials are compared. Hydrogen gas, H2, is continually bubbled into this half-cell.
a) Which one of the following would occur at the platinum electrode when the cell discharges?
A. Electrons would move from the platinum electrode through the acid solution towards the salt bridge.
B. The platinum electrode would act as the anode in this cell and have positive polarity.
C. The pH of the solution surrounding the platinum electrode would increase.
D. The hydrogen gas would be oxidised at the platinum electrode’s surface.
Solution

b) What is one change that would be expected in the Fe2+/Fe half-cell as the cell discharges?
A. Crystals of platinum would be deposited on the surface of the iron electrode.
B. The Fe2+ solution would start bubbling at the surface of the electrode.
C. Crystals of iron would be deposited on the surface of the iron electrode.
D. The Fe2+ solution would become a darker green colour.
Solution
