1) A bottle containing an unknown organic compound was examined in a university laboratory. There was an incomplete label on the bottle that gave only the empirical formula for the contents: CH4N. A chemist hypothesised that the unknown compound was 1,2-ethanediamine,
NH2CH2CH2NH2. Mass spectrometry produced the following spectral data.
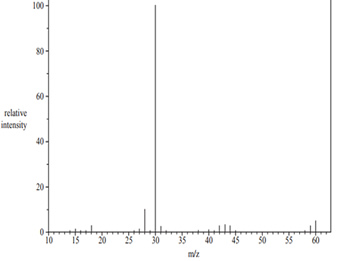
i. On the diagram above, identify the base peak.
Solution

ii. At what m/z ratio is the principal peak that supports the chemist’s hypothesis that the unknown compound has the formula
NH2CH2CH2NH2? Justify your answer.
Solution
iii. Write the semi-structural formula of the species that produces the peak at 30 m/z
Solution
Infrared (IR) spectroscopy was also used to analyse the sample. The spectrum is shown below.
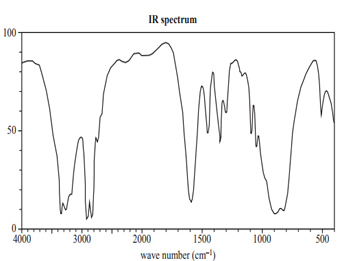
Is this spectrum consistent with the unknown compound being
NH2CH2CH2NH2? Use evidence from the IR spectrum in your response.
Solution

The sample was also analysed using 13C NMR. The spectrum is shown below.
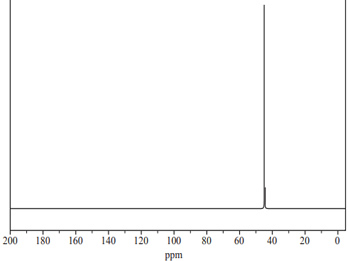
Is the 13C NMR spectrum consistent with the structure of
NH2CH2CH2NH2? Justify your answer.
Solution
