1) An unknown organic compound has a molecular formula of C4H8O. The compound is non-cyclic and contains a double bond. The infra-red (IR) spectrum of the molecule is shown below
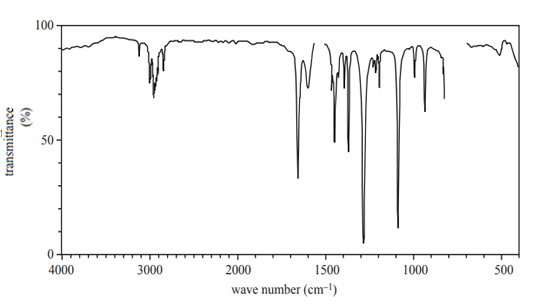
a. What does the region 3100–4000 cm-1 indicate about the bonds in
C4H8O? Give your reasoning.
.
Solution

b. The 13C NMR spectrum of the unknown compound has four distinct peaks. Draw two possible structural formulas of the unknown compound using the information provided.
Solution

c. The high-resolution 1H NMR spectrum of the unknown compound has three single peaks, as shown below.
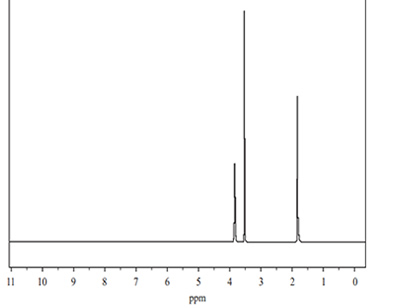
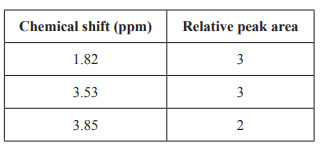
Refer to the 1H NMR spectrum and the table of spectrum information provided above. Identify three pieces of information about the unknown compound and indicate how each would assist in determining its structure.
Solution

d. C3H6O can exist as a ketone or as a primary alcohol. Explain how the principles of IR spectroscopy and 1H NMR spectroscopy lead to different spectra for the ketone and primary alcohol isomers of C3H6O, which can then be used to differentiate between the two molecules.