1. Aspartame is an ingredient in some soft drinks. Aspartame is unstable in some conditions and reacts to form four main products. One of the products of aspartame breakdown is 5-benzyl-3,6-dioxo-2-piperazineacetic acid (DKP). It is thought that DKP may be harmful to humans. A student, Kim, investigates the effect of storage temperature on the rate of production of DKP from aspartame in lemonade. Experimental data is obtained using high-performance liquid chromatography (HPLC) to analyse the aspartame and DKP content in lemonade samples.
HPLC calibration Kim first calibrated the HPLC using the following method:
1. Prepare and refrigerate a standard solution of pure aspartame with a concentration of 1000 mg L−1.
2. Transfer a 10.00 mL aliquot of the pure aspartame solution into a 1.000 L volumetric flask.
3. Fill the volumetric flask up to the 1.000 L mark with deionised water and shake the flask.
4. Inject a sample of the diluted aspartame solution into the HPLC to obtain a chromatogram.
5. Repeat steps 1– 4 with DKP. The following two calibration chromatograms were obtained
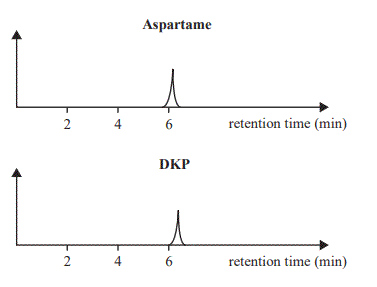
Analysis of lemonade samples
Kim then followed the method given in steps 6–14 to investigate the rate of production of DKP from aspartame in lemonade at different storage temperatures.
6. Open a can of lemonade.
7. Transfer a 10.00 mL aliquot of lemonade from the can into a 1.000 L volumetric flask.
8. Fill the volumetric flask up to the 1.000 L mark with deionised water and shake the flask.
9. Inject a sample of the diluted lemonade into the HPLC using the same operating conditions used during calibration.
10. Set up three water baths at temperatures of 15 °C, 25 °C and 35 °C.
11. Put three unopened cans of lemonade into each of the three water baths.
12. After one day, take one can from each water bath and follow steps 6–9.
13. After two days, take one can from each water bath and follow steps 6–9.
14. After three days, take one can from each water bath and follow steps 6–9.
One of the chromatograms from the diluted lemonade is given below.
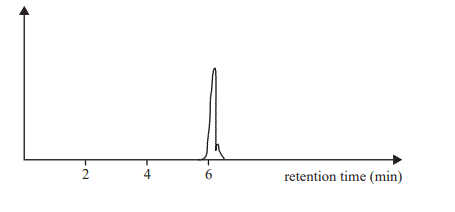
a. Using your knowledge of food chemistry, explain why aspartame is sometimes added to lemonade.
b. What is the dependent variable?
c. What steps, in addition to steps 1–14, need to be taken to use the HPLC data to measure the dependent variable? (3 marks)
Solution

d i. State a change to the operating conditions of the HPLC that could be made to reduce the errors in measuring the concentrations of aspartame and DKP.
ii. State how this change would reduce the measurement errors.
Solution

e. Kim found that the can of lemonade tested at the beginning of the experiment contained:
• 0.00178 M aspartame
• 0.00045 M DKP.
Kim quantified the remaining data from the HPLC and prepared the following table
.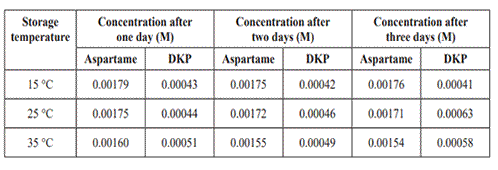
i. Write a conclusion based on the results given in the table above.
1 mark
ii. Identify a variable that has not been controlled. 1 mark
iii.
. Explain how the variable identified in part ii. affects the validity of the experiment.
Solution
