1) A student wrote the partial experimental report, shown on the right.
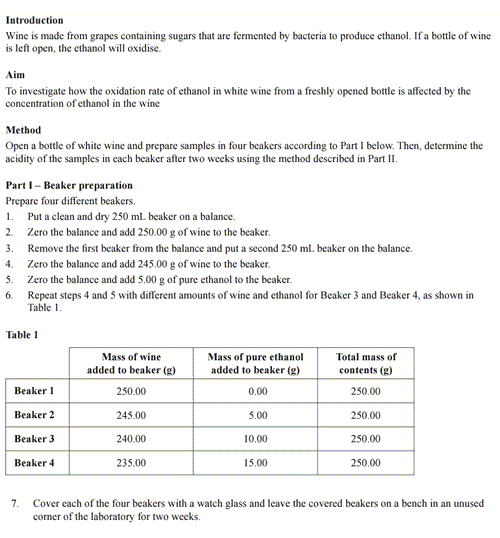

a. State how the precision of the experimental data will be affected by the acid-base titration method specified in
Part II. ? 1 mark
Solution
Immediately after the bottle was opened, an experienced and qualified laboratory technician analysed a sample of the wine and found that:
• the wine contained 8.12% m/m ethanol
• the concentration of acid in the sample was 9.45 × 10-4 M (assuming a monoprotic acid).
b. Use the laboratory technician’s results and the information given in Table 1 to calculate the initial ethanol concentration of the ethanol–wine mixture in Beaker 2 in % m/m. 3 marks
Solution
c. What is the independent variable in this investigation?
1 mark
Solution
d. At Step 5 of Part II, the student mistakenly rinsed the pipette with distilled water instead of a small amount of the contents of the beaker. Explain how this change would affect the student’s calculated value for the concentration of a monoprotic acid in the sample. 2 marks
Solution
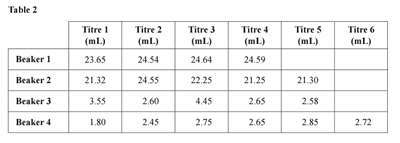
e. Write a conclusion relating to the oxidation rate of ethanol, which is consistent with the student’s results in
Table 2. 1 mark
Solution
f. Discuss your conclusion in relation to the white wine tested in this experiment. In your answer, make two points relating to one or both of the following:
• the limitations of your conclusion
• an explanation for the student’s findings
2 marks
Solution
