1) Which one of the following diagrams shows the common design features of a fuel cell?
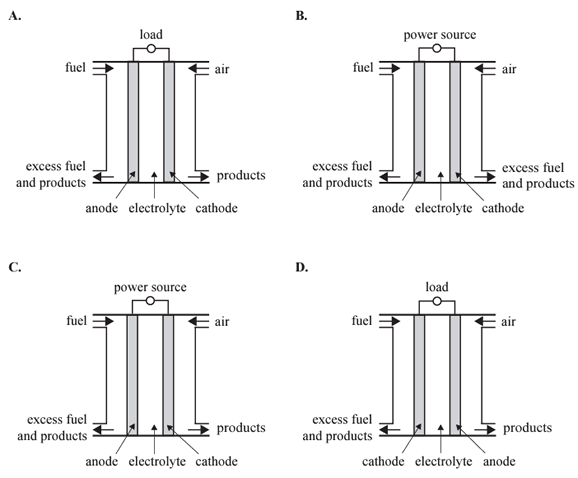
Solution

2) Galvanic cells and fuel cells have
A. the same energy transformations and both are reversible.
B. the same energy transformations and both produce heat.
C. different energy transformations but galvanic cells produce electricity.
D. different energy transformations but fuel cells use porous electrodes.
Solution

3) Unlike direct combustion of fuel, fuel cells
A. can be recharged.
B. do not produce greenhouse gases.
C. require electrical energy to overcome the activation energy barrier.
D. do not have direct contact between the oxidising and reducing agents.
Solution

4) A diagram of an acidic LPG fuel cell is shown below.
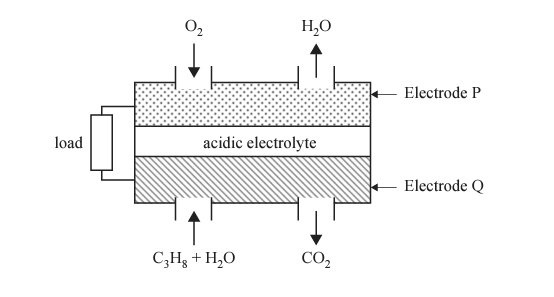
a. State the polarity of Electrode P.
b. Write a balanced half-equation for the reaction at Electrode Q.
Solution

c. Identify one difference between an acidic LPG fuel cell and a secondary cell during discharge.
Solution
