1. Which one of the following is the percentage atom economy of the reaction that converts propene to propane?
A. 91%
B. 93%
C. 95%
D. 100%
Solution

2. The correct IUPAC name for CH3CH2CHClCHOHCH3 is
A. 3-chloropentan-4-ol
B. 3-chloropentan-2-ol
C. 2,3-chloro-pentanol
D. 3,2-chloro-pentanol
Solution
3. Which one of the following chemical compounds contains a chiral carbon centre?
A. glycine
B. glycerol C.
butan-2-o1
D. 1,1-dichloropropane
Solution

4. Which one of the following reactions has the highest atom economy in the production of an organic molecule?
A. complete combustion of propyne, C3H4
B. reaction of iodine, I2 , with propane, C3H8
C. reaction of bromine, Br2, and propene, C3H6
D. formation of a dipeptide from alanine, C3H7NO2
Solution

5. A reaction pathway to produce a primary alcohol is shown below.
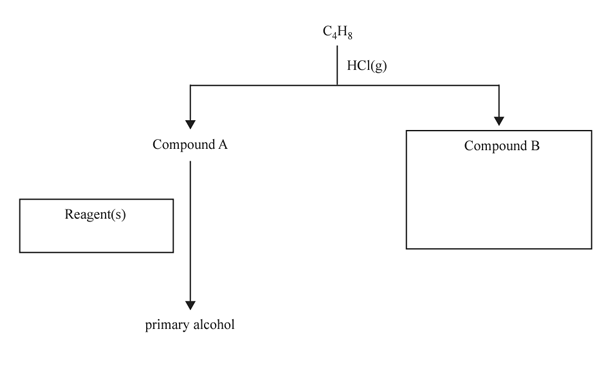
a. C4H8 reacts with HCl(g) to form two unbranched isomers – Compound A and Compound B. Only Compound A can react to produce a primary alcohol. a. Identify the type of reaction that converts C4H8 into Compound A. 1 mark
b. Write the semi-structural formula for Compound B in the box provided. 1 mark
c. State the reagent(s) required to convert Compound A into a primary alcohol in the box provided. 1 mark
Solution

d. Propan-1-ol can react with methanoic acid to produce an organic molecule.
i. Identify the catalyst for this reaction. 1 mark
ii. Write a balanced chemical equation for the reaction. 2 marks
iii. Write the systematic IUPAC name for the organic molecule produced. 1 mark
Solution
