1. A chemist uses spectroscopy to identify an unknown organic molecule, Molecule J, that contains chlorine. The 13C NMR spectrum of Molecule J is shown below.
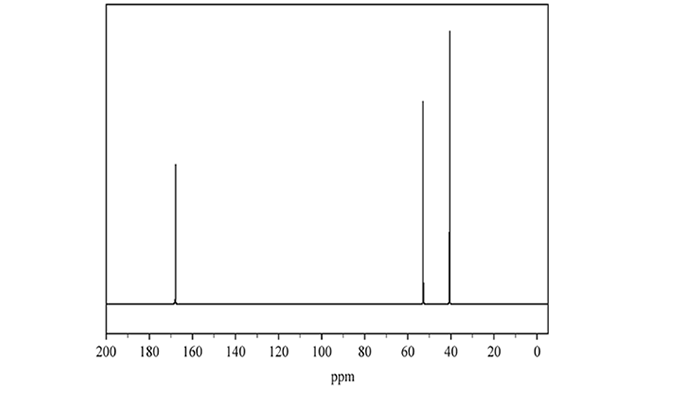
a. There are two possible carbon environments that can produce the peak at 168 ppm. Identify one of the two possible carbon environments that can produce the peak at 168 ppm.
Solution
b. The infra-red (IR) spectrum of Molecule J is shown below.
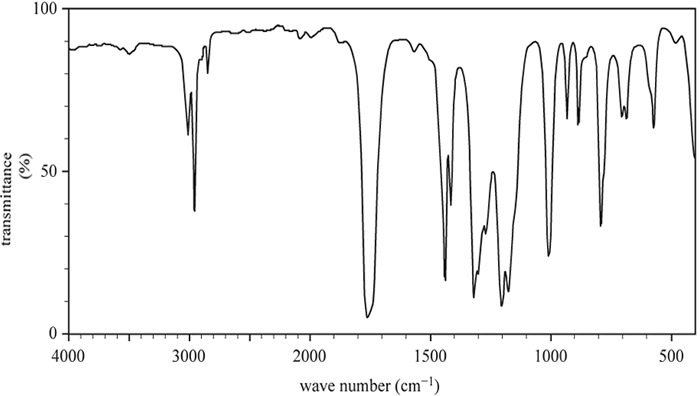
b. Name the functional group that produces the peak at 168 ppm in the 13C NMR spectrum shown above, which is consistent with the IR spectrum also shown above. Justify your answer with reference to the IR spectrum.
Solution

c. The mass spectrum of Molecule J is shown below.
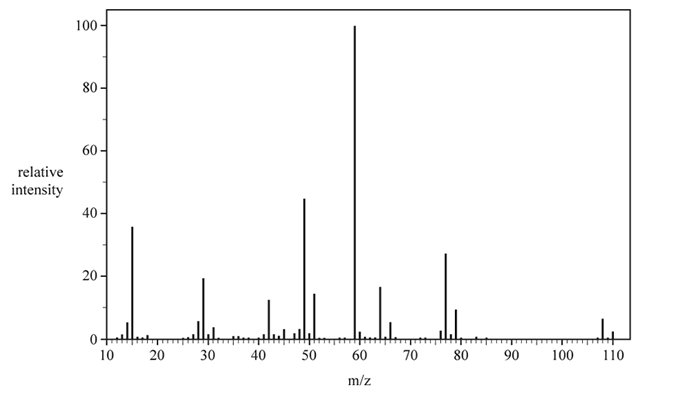
The molecular mass of Molecule J is 108.5
i. Explain the presence of the peak at 110 m/z.
ii. Explain how the peaks in the mass spectrum relate to the molecular mass of Molecule J.
Solution

d. The 1H NMR spectrum of Molecule J is shown below.
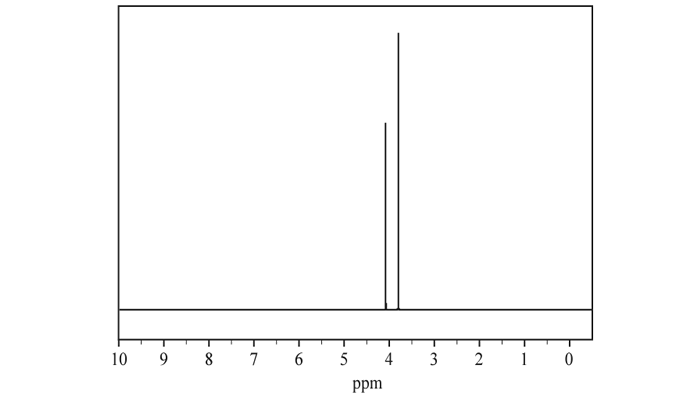
The 1H NMR spectrum consists of two singlet peaks. What information does this give about the molecule?
Solution

e. Draw a structural formula for Molecule J that is consistent with the information provided in parts a.–d.
Solution
