Chemical vs physical
During a physical change the characteristics of a substance change but nothing new is formed. During a chemical reaction however, new products are formed. Energy may also be given off in a chemical reaction or may even be absorbed (as in the case of a Cold Pack used for sports injuries). Chemical reactions are also irreversible. Irreversible means that the products can not easily be converted back to the chemicals that formed them. To reverse a chemical reaction often requires another chemical reaction to take place or the input of energy.
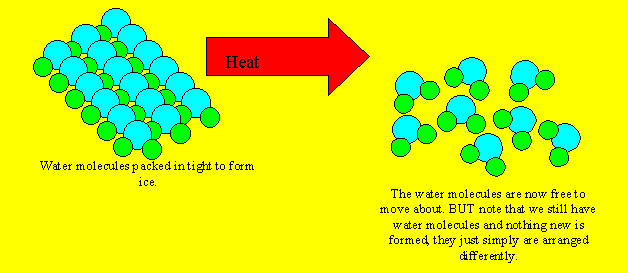
An example of a physical reaction is the melting of ice. As ice melts
into water nothing new is formed.
Other changes that can be classified as physical rather than chemical
include the:
- dissolving of sugar in water.
- heating of an iron bar until it glows red hot.
- magnetising a piece of iron
- glowing of a filament in a light globe.
- melting of an icy-pole.
- drying of wet clothes left hanging outside on hot day.
In all the above changes nothing new is formed. When sugar dissolves in
water we still have sugar but the particles have been separated and surrounded
by water molecules. The characteristics of the sugar have changed from
a crystaline, solid structure to one where all the sugar particles are
free to float around the water. Heating an iron bar does not create any
new products. The characteristics of the iron have changed so that it
glows red due to the heat energy provided. Magnetising a piece of iron
once again does not produce a new substance.but changes the characteristics
of the iron. Iron is still present but it is now magnetised. The glow
of a lamp filament is due to the electrical energy that is provided. Melting
of an icy-pole is a change in the characterisitcs of the icy-pole. It
changes from a solid to a liquid. When wet clothes are left to dry, water
changes states from liquid to a gas. No new product is formed.
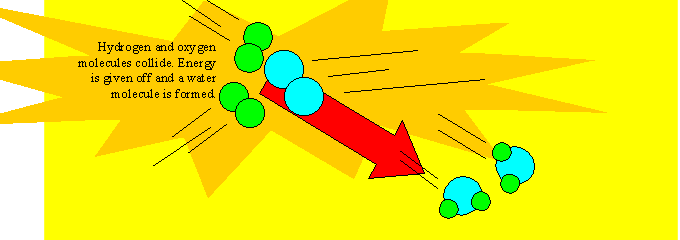
An example of a chemical reaction is the burning of hydrogen gas and oxygen gas to form water. Water is formed from the reaction between hydrogen and oxygen and like all chemical reactions this process is not easily reversed.