Measuring the rate of a chemical reaction
Effect of concentration
Aim - to measure the effect of concentration of reactants on the rate of reaction.
Method - using a measuring cylinder, pour 20ml of 2MHCl into a 50ml beaker. Weigh approximately 20 grams of marble(calcium carbonate)chips into a second 50ml beaker and weigh. Place both beakers on an electronic balance. Record the reading on the balance.
Quickly pour the acid into the beaker with the marble chips. Place the empty beaker back on the balance. Record the readings every 15 seconds. Continue until no further change in mass is observed. Record the results in a table form.
Plot a graph of the mass loss against time. Repeat the above procedure using acid solution of 0.1M concentration.
Record your results in table form and plot a graph of mass loss versus time.

Click to see a 120kb video
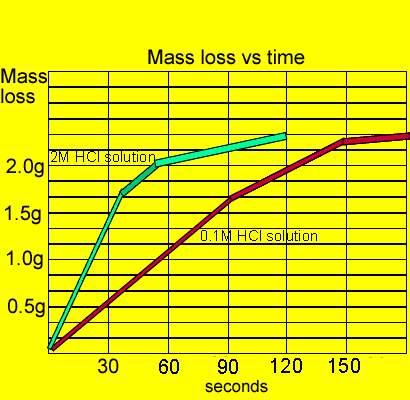
The results shown here are not actual results and are designed to assist students visualise the experiment prior to attempting it in class.
Green line represents 2MHCl solution while the red represents 0.1MHCl solution.
Questions
1) The reaction between calcium carbonate and hydrochloric acid is represented
by the equation below.
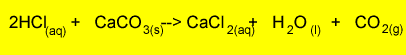
How do you account for the mass loss measured?
2) Plot your results on a graph and explain in terms of the rate of collisions
why the graphs look as they do.