Rates of reaction
Inorganic Catalysts
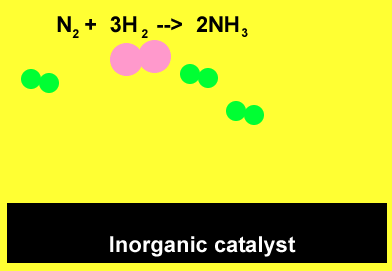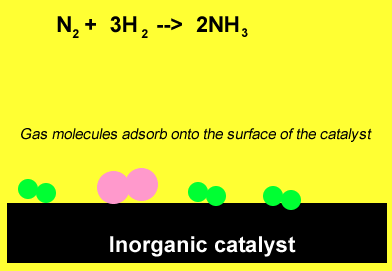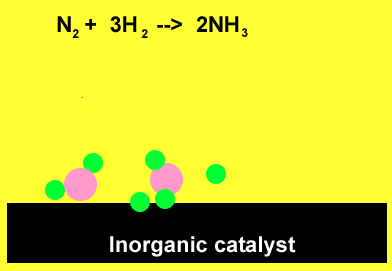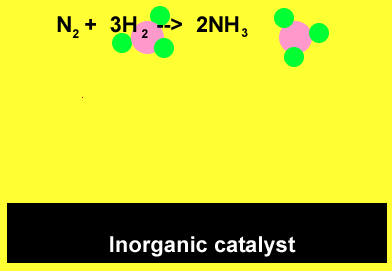
The animation above demonstrates the ability of the surface of inorganic catalysts, at relatively low temperatures, to break chemical bonds. This results from the presence of a high surface energy. The individual atoms quickly recombine to form ammonia.
In the absence of a catalyst temperatures up around 3,000 oC are required to cause the reactant molecules to collide with enough force as to break chemical bonds.