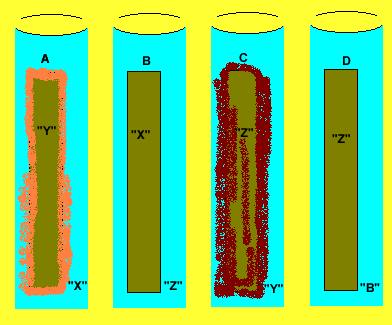
A student set up the experiment above.
Test tube "A" contains metal "Y" in a solution containing
metal "X".
Test tube "B" contains metal "X" in a solution containing
metal "Z".
Test tube "C" contains metal "Z" in a solution containing
metal "Y".
Test tube "D" contains metal "Z" in a solution containing
metal "B".
1) Place the metals "X", "B", "Y", "Z"
in order of increasing reactivity.
2) A bridge is made of metal "Y" while the bolts used to secure the beams are made from metal "B". Explain what will happen.
3) A bridge is made of metal "Z" while the bolts used to secure the beams are made from metal "Y". Explain what will happen.
Which is the more reactive metal out of copper and iron?


Describe what happened by filling in the gaps. Select
the correct response.
*Hint keep in mind that ions are atoms that have a positive
or negative charge because they have either lost or gained electrons.
Metals usually form positive charged ions.
The blue copper sulfate solution contained
. Iron is
than
copper so when the two metals react, are transferred from the
atom to the ion.
The blue copper sulfate solution contained copper ions. Iron is
more reactive than
copper so when the two metals react, electrons are transferred from the iron
atom to the copper ion.
Hide solution
Explain the results.