Electrolysis with solutions
Electroplating
Electroplating involves the
deposition of a layer of metal on top of another metal. Often the silverware
you see on dinner tables is cheap iron coated with a thin layer of silver.
This is done by a process called electroplating in an electrolytic cell.
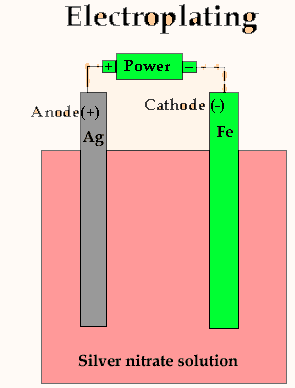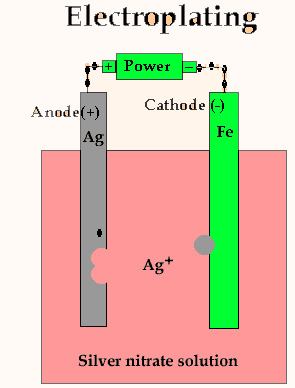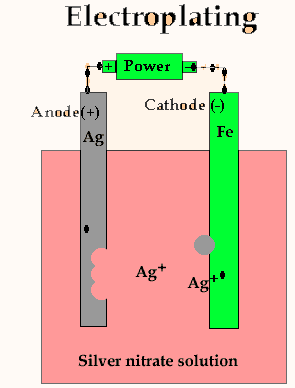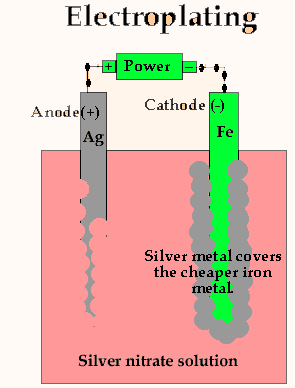
As is shown in the animation on the right, the metal we wish to coat is
placed on the negative terminal of the power source. This is then immersed
in an electrolyte that contains ions of the metal we wish to coat with,
in this case, silver ions. To the positive terminal we connect a silver
metal.
Electrons are pumped out of the silver metal in effect corroding it and releasing silver ions into the electrolyte solution.
Ag(s) => Ag+(aq)+ e
The positive ions migrate over
to the negative electrode (the metal we wish to coat) and regain their
electrons to form silver metal once again.
Ag+(aq)+ e =>Ag(s)
The ions that
migrate are not always the ones that have just being released from the
silver metal but may be any silver ion in the electrolyte. The ions that
are released from the silver metal just replace the ions that have been
taken out of the solution. A constant silver ion concentration is maintained.
Give the polarity
and the reaction that occurs at the cathode.
Give the polarity and the reaction that occurs at the anode.
Electroplating is not as simple as the animation above shows. If we were to coat a piece of iron in this manner a loose black sludge would develop. In practice the voltage, current, temperature and electrolyte are carefully controlled.
Looking at the animation above it is easy to see the relationship between number of atoms of silver deposited and the number of electrons that pass through the circuit.
This relationship is very important and leads us into the next chapter where we can calculate the mass of metal deposited given the current supplied.