Solution
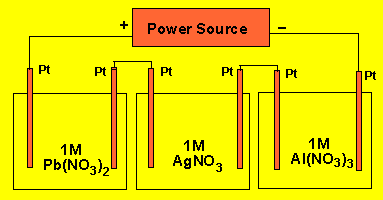
Three cells are connected as
shown below. A steady current is passed through the system for a fixed length
of time. If 2.14 grams of silver is deposited at the electrode in the centre
cell what mass of metal is deposited in each of the other cells?
Atomic mass of Ag = 108, Pb
= 207, Al = 27
Step 1 Calculate
the mole of silver metal deposited.
2.14/107 = 0.02
mole of silver metal
Step 2 Calculate
the number of mole of electrons passed through the system in order to deposit
0.02 mole of silver.
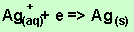
According to the equation above
1 mole of electrons produces one mole of silver metal. To produce 0.02 mole
of silver we need 0.02 mole of electrons.
Step 3 Calculate
the mole of Pb deposited if 0.02 mole of electrons was passed through the
system
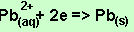
According to the
equation above for 0.02 mole of electrons 0.01 mole of Pb is deposited.
The mass of Pb = 0.01 X 207 =2.07 grams.
Aluminium can
not be deposited from an aqueous solution containing aluminium ions.
Click
to hide the solution
Solution
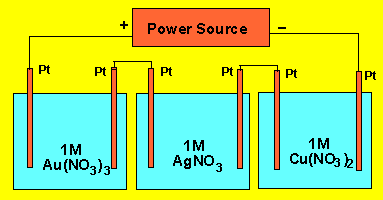
Irene sets up a circuit for electroplating
silver, copper and gold in three separate cells. The cells are connected in
series as shown on the right. A current flows for 30 minutes and 2.58 grams
of copper is deposited. Calculate the masses of silver and gold deposited.
Step 1 Calculate the
mole of copper atoms deposited.
2.58 / 63.54 =0.041
mole
Step 2 Calculate the
mole of electrons passed through the system
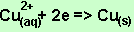
According to the equation above,
0.041 mole of copper deposit from 0.082 mole of electrons
Step 3 Calculate the
mass of Au formed
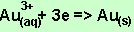
According to the
equation above for 0.082 mole of electrons 0.082/3 mole of Au is deposited.
The mass of Au deposited is 0.02/3 X 196.97 =5.38 grams
Step 4 Calculate the
mass of Ag formed
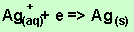
According to the
equation above 0.082 mole of electrons produce 0.082 mole of silver.
The mass of silver is 0.082 X 107.87 =8.85 grams
Click
to hide the solution
Solution
Irene sets up a circuit for electroplating
silver, copper and gold in three separate cells. The cells are connected in
series as shown on the right. A current flows for 30 minutes and 2.58 grams
of copper is deposited. Calculate the current used.
Atomic masses Cu =63.54, Ag =107.87,
Au = 196.97
Step 1 Caluclate the mole of copper
atoms deposited
2.58/63.54 =0.041
mole
Step 2 Calculate the
mole of electrons passed through the system
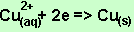
According to the
equation above if 0.041 mole of copper is deposited 0.082 mole of electrons
pass through.
Step 3 Calculate the
charge from 0.082 mole of electrons
0.082 X 96,500 =
79,130C
Step 4 Calculate the
current needed
Q =I X t
79,130 = I X 30 X 60
4.40A = I
Click
to hide the solution
Solution
Jonathon sets up a circuit for
electroplating silver, copper and gold in three separate cells. The cells
are connected in series as shown on the right. A current flows and 4.78 grams
of copper is deposited. Calculate the masses of silver and gold deposited.
Atomic masses Cu =63.54, Ag =107.87,
Au = 196.97

Step 1 Calculate the
mole of copper atoms deposited.
4.78 / 63.54 =0.075
mole
Step 2 Calculate the
mole of electrons passed through the system
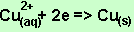
According to the equation above,
0.075 mole of copper deposit from 0.15 mole of electrons
Step 3 Calculate the
mass of Au formed
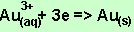
According to the
equation above for 0.15 mole of electrons 0.15/3 mole of Au is deposited.
The mass of Au deposited is 0.15/3 X 196.97 =9.88 grams
Step 4 Calculate the
mass of Ag formed
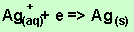
According to the
equation above 0.15 mole of electrons produce 0.15 mole of silver.
The mass of silver is 0.15 X 107.87 =16.18 grams
Click
to hide the solution
Electrolysis
in solutions exercises 3
|
1)
Three cells are connected as shown on the right. A steady current is passed
through the system for a fixed length of time. If 2.14 grams of silver is
deposited at the electrode in the centre cell what mass of metal is deposited
in each of the other cells?
Atomic mass of Ag = 108,
Pb = 207, Al = 27
Solution
|
 |
|
2) Irene sets up a circuit
for electroplating silver, copper and gold in three separate cells. The
cells are connected in series as shown on the right. A current flows for
30.0 minutes and 2.58 grams of copper is deposited. Calculate the masses
of silver and gold deposited.
Atomic masses Cu =63.54, Ag
=107.87, Au = 196.97
Solution
|
 |
3) What is the
current used in question 2) above?
Solution
|
|
4) Jonathon sets up a circuit
for electroplating silver, copper and gold in three separate cells. The
cells are connected in series as shown on the right. A current flows and
4.78 grams of copper is deposited. Calculate the masses of silver and
gold deposited.
Atomic masses Cu =63.54, Ag
=107.87, Au = 196.97
Solution
|
 |

![]()
![]()
