Building a half
reaction
in an acid solution
Some easy steps to follow.
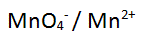
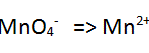
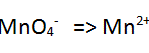
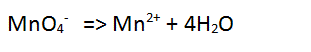
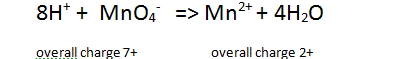
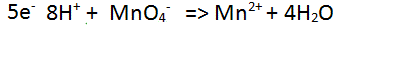

Continue
to see how the half reactions are used to form the overall cell reaction.
|
Building a half
reaction Some easy steps to follow. |
|
| The permanganate ion is reduced to the manganese 2+ ion |
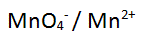 |
| Step 1 Write the two species on either side of an equation |
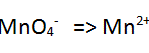 |
| Step 2 Balance all the atoms of the half equation except for oxygen and hydrogen. In this case they are balanced. |
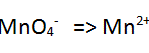 |
| Step 3 Balance the oxygen atoms by adding water to the side deficient in oxygen atoms. |
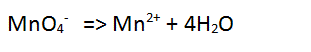 |
| Step 4 Balance for hydrogens by adding hydrogen ions to the side deficient in in hydrogens |
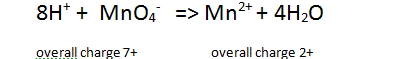 |
| Step 5 Balance the charge on each side of the equation by adding electrons to the most positive side. |
|
| Step 6 Place the states of reactants and products. |  |
| Continue with half cell reactions in an alkaline solution. | |
|
Continue
to see how the half reactions are used to form the overall cell reaction.
|
|