Constructing
an electrochemical cell
from an overall equation
We can construct an electrochemical cell from an overall equation. We must follow a few steps as outlined below.
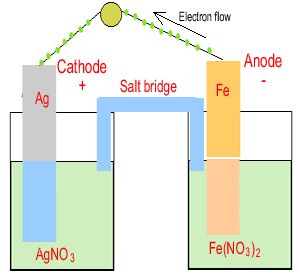
For example. Iron metal is placed in a solution of silver nitrate and reacts according to the equation below.
Fe(s) + 2Ag+(aq) => 2Ag(s) + Fe2+(aq)
Step 1 Obtain the two half equations.
Reduction - Ag+(aq) + e => Ag(s)
Oxidation - Fe(s) => Fe2+(aq) + 2e
Step 2 Construct the following
half cells
Ag+(aq) / Ag(s) and Fe(s)
/ Fe2+(aq)
If a metal is present in the half cell use it as the electrode. Otherwise use inert carbon or platinum rods.
1) Construct an eletrochemical cell from the overall equation given below.
Cu(s) + 2Ag+(aq) => 2Ag(s) + Cu2+(aq)
5Cu(s) + 2MnO4-(aq) + 16H+(aq) => 2Mn2+(aq) + 5Cu2+(aq) + 8H2O(l)