Fuel cells have been around for many years and were widely used in the space program during the 1960's. A fuel cell used to provide electricity for the Apollo space program is shown on the right.
Fuel cells vary from other types of cells in that the reactants are supplied continuously and the products continuously removed allowing for an indefinite supply of electricity.
Electrodes have three distinct properties:
-the obvious one is that they must conduct electricity;
-they must be porous to allow contact between gases and electrolyte;
-act as catalysts
.

A fuel cell has three main parts:
- a membrane;
- an anode catalyst ;
- a cathode catalyst.
Hydrogen is fed into the cell and flows over the anode catalyst where the molecule separates into two hydrogen ions(protons) releasing two electrons. The electrons and protons flow through the cell via separate pathways. The electrons flow through an external circuit while the protons flow through a special membrane that allows only positive ions to flow through it. On the other side, at the cathode catalyst, the oxygen molecule is separated into two oxygen atoms. As electrons arrive, the oxygen atoms react with the hydrogen ions forming water.
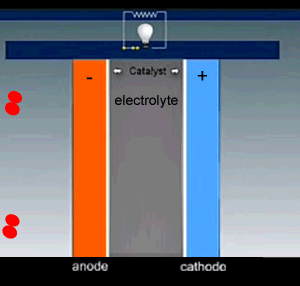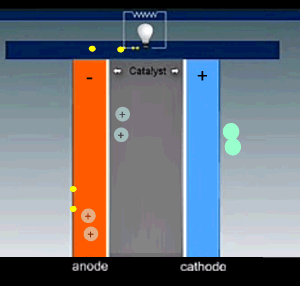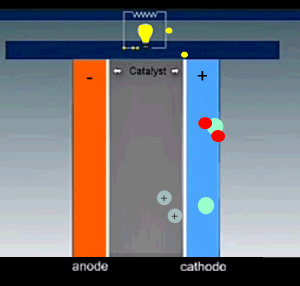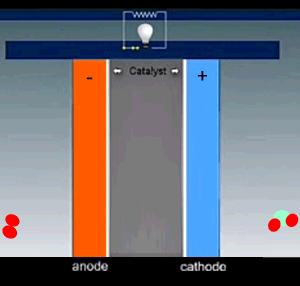
The nature of the electrodes is critical for the proper function of the fuel cell as they act as catalysts and the current depends on their surface area.
The reactions are :
- at the anode - H2(g) => 2H+(aq) + 2e-
- at the cathode O2(aq) + 4H+(aq) + 4e- => 2H2O(l)
The overall reaction can be summarised as
O2(aq) + 2H2(g)=> 2H2O(l)
Characteristics of a fuel cell.
- Fuel cells are about 70% efficient in converting chemical energy into electrical energy as compared to 30% efficiency for coal and gas powered electrical generators.
- Reactants are supplied continuously and products are removed.
- Reactants are not limited to just H2 and O2 and include CH4, CH3OH and C3H8
- Electrodes are porous and act as catalysts
- Since reactants are constantly supplied the voltage is relatively constant unlike galvanic cells whose voltage output decreases as reactants are depleted and products build up.
- Because reactants have to be fed constantly into the fuel cell and products removed they can be cumbersome and hard to transport requiring very expensive high pressure and low temperature equipment.
Not all fuel cells operate on hydrogen and oxygen . Some small fuel cells use ethanol to react it with oxygen to produce ethanoic acid and water to produce a small output of 1.2V. The overall reaction is shown below.
-
CH3CH2OH(l) + O2(g) ==> CH3COOH(aq) + H2O(l)
-
The half-cell reactions are:
-
(ii) Reduction, +ve electrode: O2(g) + 4H+(aq) + 4e- ==> 2H2O(l)
-
(iii) Oxidation, -ve electrode: CH3CH2OH(l) + H2O(l) ==> CH3COOH(aq) + 4H+(aq) + 4e-
-
Give the reaction occurring at the anode.
Give the reaction occurring a the cathode.
Give the overall reaction
Give the reaction occurring at the anode.
Give the reaction occurring a the cathode.
Give the overall reaction
The hydrogen fuel cell is one of the oldest fuels cells, used in the Apollo Space program. In the hydrogen fuel cell, hydrogen gas reacts with oxygen gas according to the equation below.
2H2(g) + O2(g) => 2H2O (l)
This cell is also called the alkaline fuel cell because it operates with an alkaline electrolyte.
Give the reaction that occurs at the anode.
Give the
reaction that occurs at the cathode.
Solution