When considering redox reactions involving organic compounds it is often confusing assigning oxidation numbers to carbon. For example consider the combustion reaction below.
CH3CH2CH3(g) + O2(g) => CO2(g) + H2O(g)
Is this a redox reaction? Well it appears to be. Obviously propane is oxidised because oxygen is added and oxygen is reduced from 0 to -2. But it is difficult to see which element in propane is oxidised.
What is the oxidation state of carbon in propane?
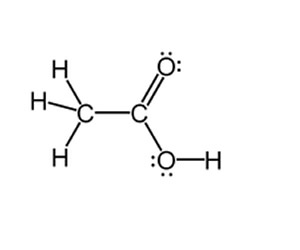
When assigning oxidation states to carbon in organic compounds such as ethanoic acid, pictured on the right, we treat each carbon separately.
For example the CH3 group is treated separately to the COOH group.
We ignore the c-c bond and treat the CH3 individually.
C + 3 X 1 = 0
=> C = -3
In a similar way we treat the COOH group.
C + 2X -2 + 1 = 0
=> C = +3
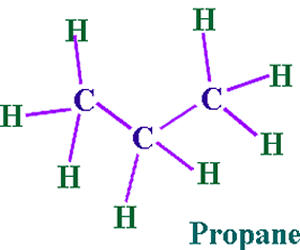
Let's take propane for example.
We have two CH3 groups and one CH2 group.
Carbon has two oxidation states in propane.
- +3 in CH3 and -2 in CH2.
Calulate the oxidation state/s of carbon in the following compounds.
a) CH3OH
b) HCHO
c) CCl4
d) CH3COO-
e) CH3CH2COOH
Solution
