Before we look at the charging and recharging of secondary cells, let's look at a battery using a simple water analogy.
A battery can be considered as having a store of electrons at high voltage that it can push around a circuit and capture them back in its low voltage reservoir. Much like water at high pressure can be pushed around a circuit to a point of low pressure, as shown in the animation on the left.
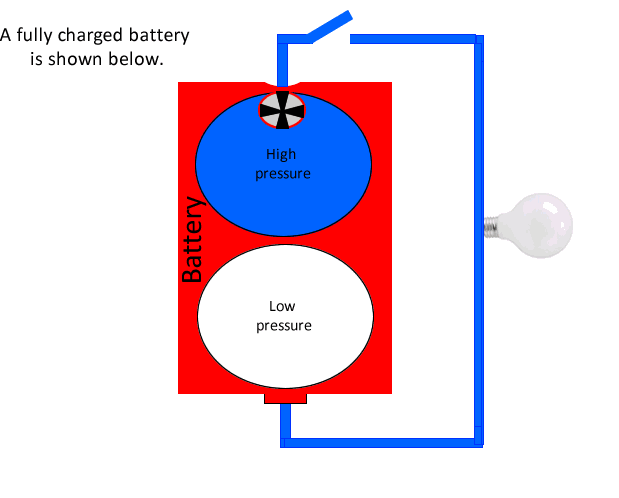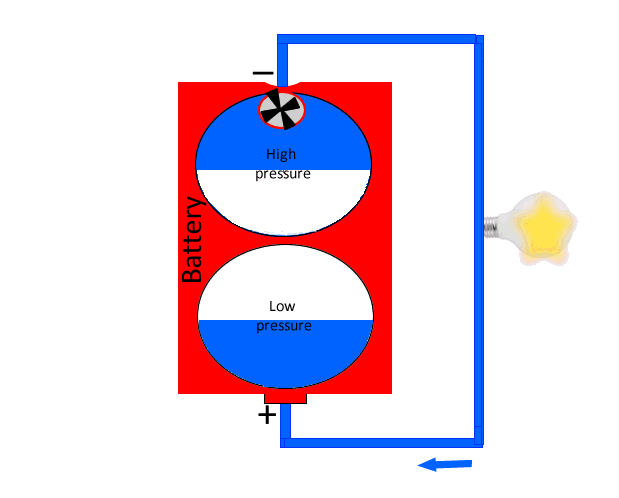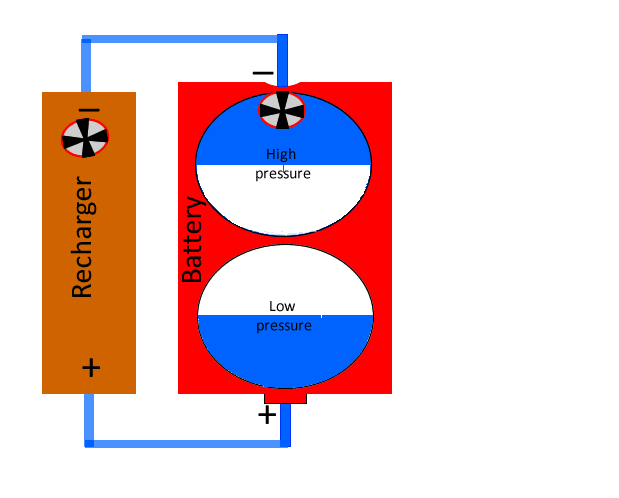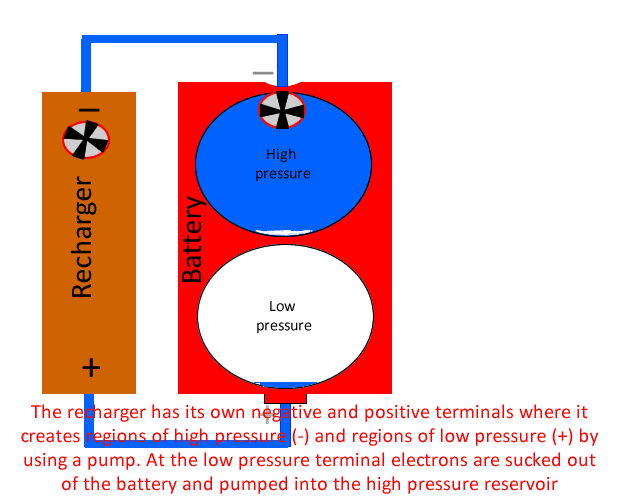
When the battery is running low a recharger can be used to pump electrons out of the low voltage area to the high voltage area of the battery. Using our water analogy the recharger can be considered a pump that uses energy to pump water from the low pressure area of the battery to the high pressure area.
The negative terminal of the battery represents the area of high voltage or high pressure as for our water analogy, while the positive terminal is the low pressure reservoir.
Now when we apply the pump or recharger to the battery the pump has a terminal which is also positive because at its positive terminal it creates a low pressure environment to literally suck the water (electrons) out of the low pressure reservoir and pump them, using energy into the high pressure reservoir.
That is why the positive terminal of the recharger is connected to the positive terminal of the battery and the negative terminal of the recharger is connected to the negative terminal of the battery.
Batteries are generally portable devices that convert chemical potential energy into useful electrical energy. The overall chemical reaction that takes place is divided into two chemical processes that are physically separate:
-
at the negative terminal, during discharging, the process of oxidation takes place, where the charge of one of the reactants becomes more positive;
- at the positive terminal, during discharge, the process of reduction takes place where the charge of one of the reactants becomes more negative.
Click for more detail on the lead-acid accumulator (car battery)

A battery is a device that provides two separate pathways for charge to flow between the two physically separate sites of oxidation and reduction. A simplified version of a battery is shown on the right.
One pathway involves the flow of electrons from the site of oxidation to the site of reduction through an external circuit. The other pathway provides for the flow of negative ions form the site of reduction to the site of oxidation. These ions pass through an insulated and conducting medium, electrolyte, that connects the sites of oxidation and reduction.
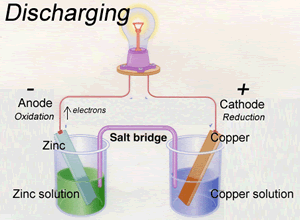
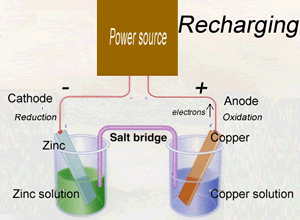
When recharging a battery the direction of electron flow is reversed. An external power source provides energy for a reduction reaction to occur at the negative electrode and an oxidation reaction to proceed at the positive electrode.
The reactions that occur at each terminal when discharging, are reversed by the supply of energy from an external energy source on recharging. In order for recharging to occur it is crucial that the products from the reactions that occur during discharging stay in contact with the electrodes.
When trying to recharge non-rechargeable battery, at least one of the oxidation-reduction reactions may not be reversible and at times the unwanted reactions take place to produce gas. This can be a dangerous situation.

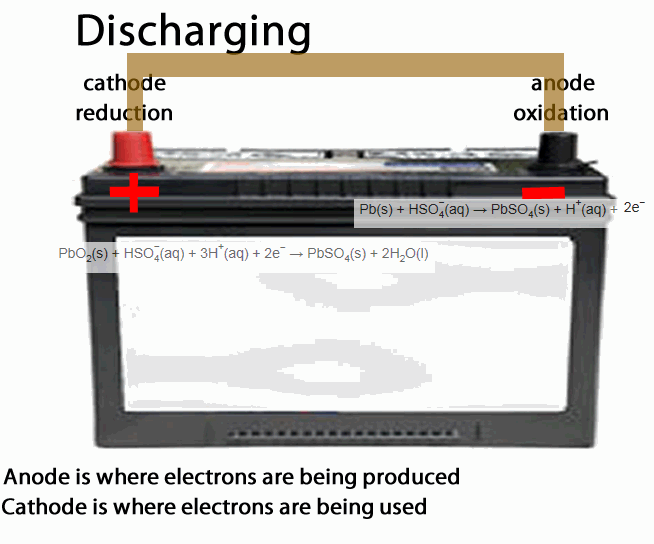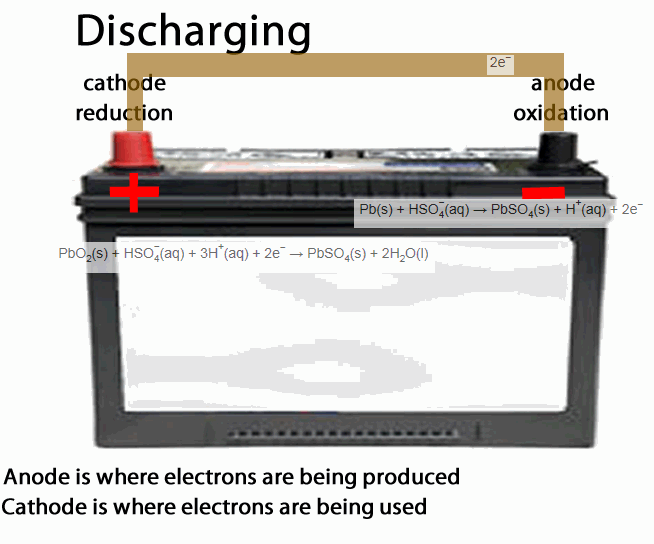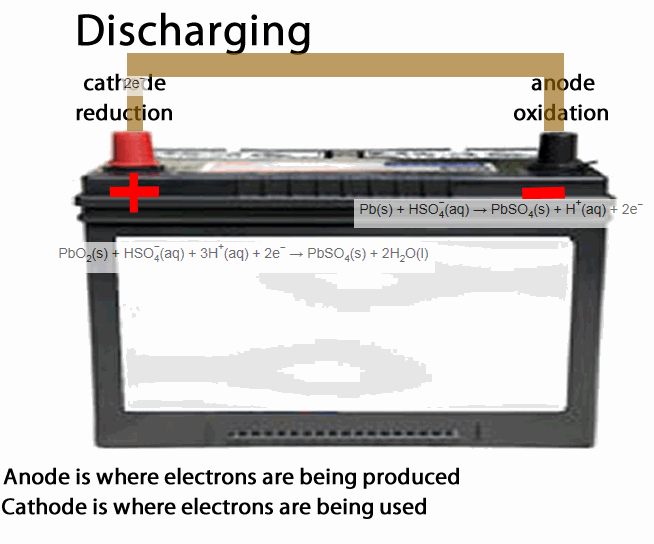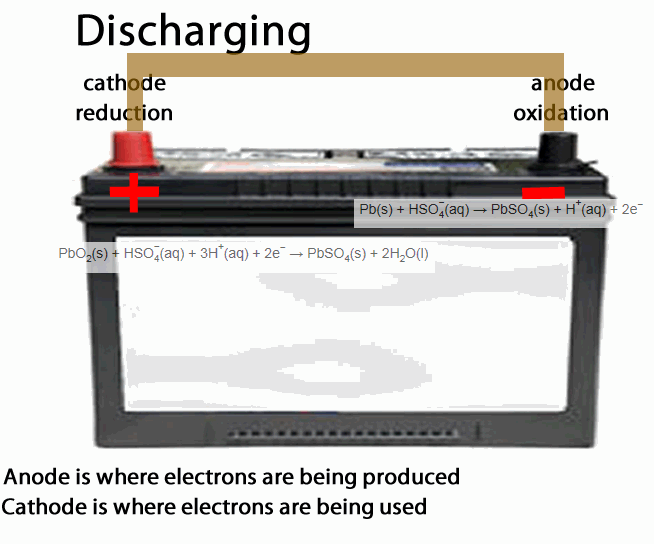
The lead acid accumulator, or the humble car battery, is a fine example of a secondary cell.
Note that in both recharging and discharging the:
- anode is the place of oxidation, where electrons come out from. During discharging the anode is negative during recharging the anode is positive
- cathode is the place where electrons go into, in other words the site of reduction. During discharging the cathode is positive during recharging the cathode is negative