Amount of substance
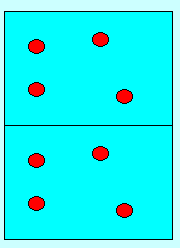
Each square on the right represents 1 litre of solution.
Each litre, as you can see, contains 4 molecules of substance "A".
The diagram shows 8 "A" particles in 2 litres so the concentration
of the solution on the right is therefore
amount/volume = 8/2 = 4"A" per litre
How many "A" particles will be present in 4litres?
How many "A" particles will be present in 6litres?
How many "A" particles will be present in 0.5litres?
BY now you it should be obvious that to calculate the number of particles
we multiply the volume by the concentration.
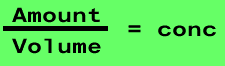
The formula for calculating concentration can be manipulated to calculate the amount of substance in a given volume.
We can now calculate the amount of substance in a given volume of solution if we know the concentration.
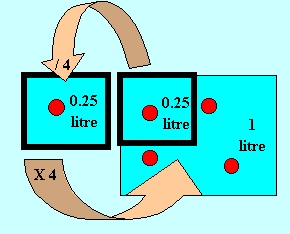
The diagram on the right shows a 1 litre solution with a concentration
of 4"A" per litre. The amount of "A" in 0.25 litres
is given by the calculation
concentration X volume =amount
4 X 0.25 = 1 "A" particle.