Solubility
supersaturated solution
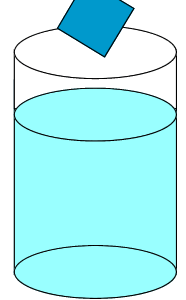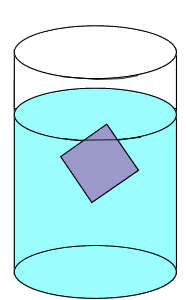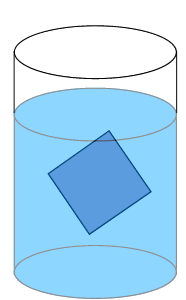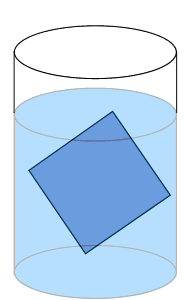
Solute particles precipitate out of solution and the rate at which particles leave the crystal is significantly lower than the rate at which solute particles stick to the crystal.
Solute particles will continue to precipitate until a saturated solution is achieved.
