A human error, or mistake, is an unintended action or omission by the person conducting the experiment. These are often random, avoidable, and result from inattention, lack of experience, or misunderstanding. They typically do not follow any consistent pattern and can vary in each trial.
Errors in Titration: May be systematic or random, arising from the experimental design, procedure, or external factors, such as temperature which may impact the volume of tirant or sample delivered. Systematic errors are consistent and reproducible, while random errors are unpredictable and vary across trials.
The following are examples of errors.
· The procedure specifies the wrong indicator: Using phenolphthalein will cause overshooting of the endpoint thus leading to a calculated concentration that is higher than is actually is in the sample.
· Parallax Error: When reading the volume on the burette, if the observer’s eye is not level with the meniscus, a parallax error can occur. This can cause the recorded volume to be slightly higher or lower than the actual volume, leading to inaccurate titration results. If the observer’s eye is constantly positioned at the exact spot every reading then it becomes a systematic error, which cancels when taking the difference between two readings of a burette.
· Side reactions which may react with the titrant – The presence of a secondary acid in the sample mixture that reacts with the titrant (base).
·The method specifies the use of a base that is not a primary standard, eg NaOH.
Some mistakes include:
· Incorrect use of equipment. For example:
- Not filling the volumetric flask to the mark by overshooting or undershooting the mark.
- Inaccurate measurement of reagents when the experimenter fails to measure the volumes accurately, such as by not properly reading the meniscus in the pipette or burette.
· Endpoint Determination: Misjudging the endpoint of the titration is a common source of error. If the colour change of methyl orange, indicating the endpoint, is not observed correctly, it can result in either overshooting or undershooting the actual endpoint, leading to inaccurate measurements of the amount of titrant used.
·Inadequate Cleaning of Apparatus: If the burette, pipette, or flask are not properly cleaned before use, residual chemicals from previous experiments could contaminate the solution, altering the reaction and resulting in errors in the titration process. If the wrong apparatus is washed with water dilution can occur
·Spilling of sample solution as it is delivered into the conical flask.
·Leaving the funnel on top of the burette while conducting the titration. These may cause titrant to drip from the funnel into the burette.
· Incomplete mixing or failure to mix the solution in the conical flask whilst titrating: During the titration process, if the solution in the flask is not thoroughly mixed after each addition of titrant, it can result in localized concentrations of the titrant, leading to an inaccurate determination of the endpoint. This can cause inconsistencies in the titration results.
· Misreading the Burette: If the person conducting the titration misreads the burette scale due to parallax error or distraction, they may record an incorrect volume of titrant, leading to an inaccurate concentration calculation.
·Parallax Error: When reading the volume on the burette, if the observer’s eye is not level with the meniscus, a parallax error can occur. This can cause the recorded volume to be slightly higher or lower than the actual volume, leading to inaccurate titration results. |
|
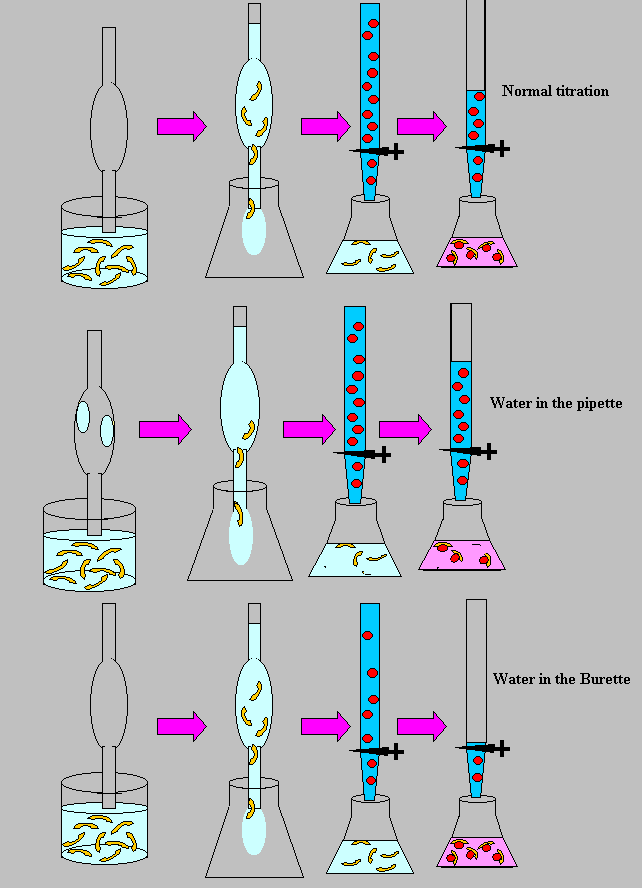
Titration as you already know is a very accurate procedure in measuring
volume of liquids. However, because it is so sensitive and accurate many
errors can occur throughout the procedure to give erroneous results. One
very common source of error is contaminated glassware (pipette, burette).
Contamination doesn't always come in the form of chemicals, water is a
contaminant in the titration procedure because it acts to dilute our solutions.
Since you are familiar with the titration
procedure, complete the table below.
20ml aliquots(samples of the unknown) of an unknown acid
solution were titrated each time with base. The average titre (volume
of base solution added from the burette) is given for a normal, error
free titration. Indicate wether the average titre will be lower or higher
than the normal.
Click on the green cell to reveal more information. Double click to hide
the information.
|
|
|
|
|
33.45
|
|
Water in the pipette (mistake, careless protocol)
|
> 33.45,< 33.45 or = 33.45
|
Water in the burette (mistake, careless protocol)
|
> 33.45,< 33.45 or = 33.45
|
|
|
> 33.45,< 33.45 or = 33.45
|
|
Acid in the burette (mistake, careless protocol)
|
> 33.45,< 33.45 or = 33.45
|
|
Acid solution in the flask (mistake, careless protocol)
|
> 33.45,< 33.45 or = 33.45
|
|
Solutions
|
|