Stoichiometry-using the ratios-part 3
Consider the problem given below.
How many mole of AlCl3 are formed if 4 mole of NaNO3 are also formed?
From the equation we see that for every 3 mole of NaNO3 formed 1 mole of AlCl3 is formed.
So we take the 4 mole of NaNO3 and multiply it by 1/3 to get the mole of AlCl3
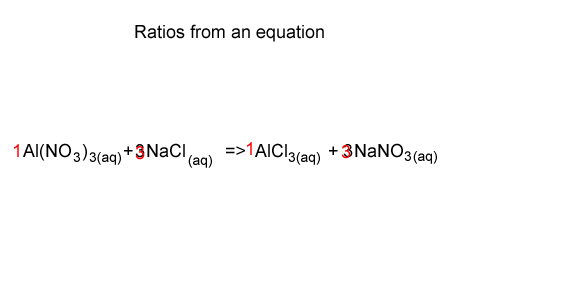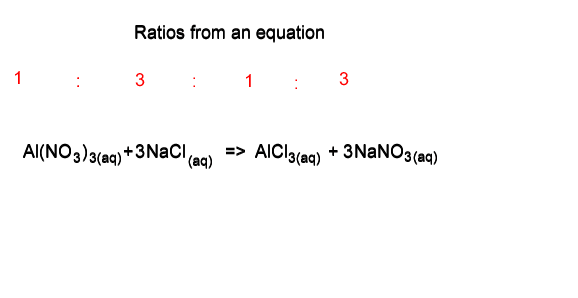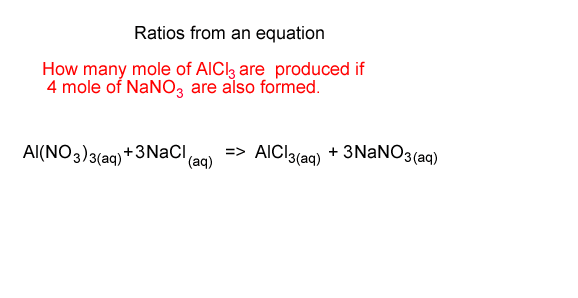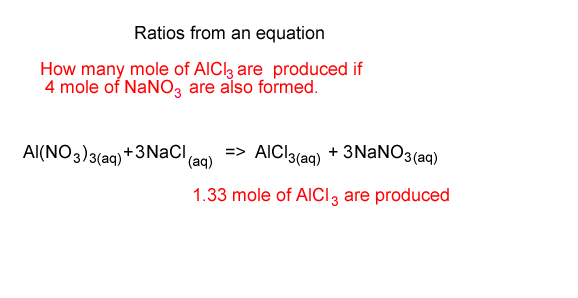
Consider the problem given below.
From the reaction:
B2H6 + 3O2 => 2HBO2 + 2H2O
What mass of O2 will be needed to burn 55.2 g of B2H6?
Play the video on the right for solution.
Consider the problem given below.
From the reaction:
B2H6 + 3O2 => 2HBO2 + 2H2O
Calculate the number of O2 molecules that are needed to produce will be needed to burn 20.5 g of H2O.
Play the video on the right for solution.