Stoichiometry
Excess (too much reactant)
| The equation pictured on the right is of a simple chemical reaction. The bolt and the nut represent elements forming the compound (nut+bolt). | 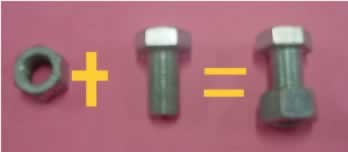
|
|
| Sometimes we can have too much of a particular reactant. In this case we have too many nuts and not enough bolts. |  |
|
. |
How much of
the compound (nut+bolt) that is formed is determined by the number
of bolts. There is obviously not enough bolts to react with all the
nuts. As soon as the bolts are used up the reaction will cease. Two
units of the compound are formed and two nuts are left unreacted. |
|
| The reactant that is present in excess amount is known as the excess reactant, while the reactant that will be used up is known as the limiting reactant | ||