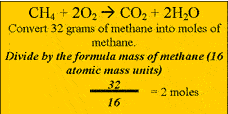
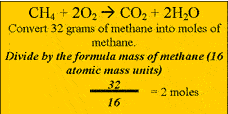
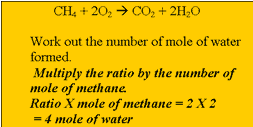

|
Mass-mass stoichiometry |
|
|
Methane burns in oxygen to produce carbon
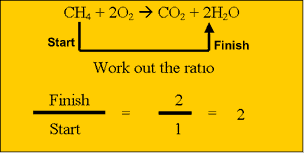
dioxide and water according to the equation above. Calculate the amount, in mol, of methane that reacts completely. Step 2 Calculate the ratio from the prefixes in the balanced equation above Step 3 Calculate the number of mole of water produced. Step 4 Calculate the mass of water produced. |