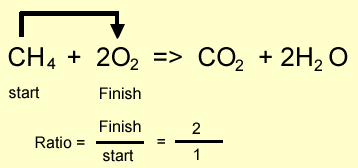
Divide the mass of methane (64 grams) by its formula mass.
Formula mass of methane is 16.
The number of moles of methane is therefore given by the expression
64/16 = 4mole
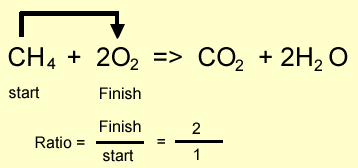
The number of mole of oxygen are calculated as shown below.
mole of methane X ratio = mole of oxygen
4 X 2 = 8 mole of oxygen
Calculate the mass of oxygen as given by the expression below.
mole X formula mass = mass
The formula mass of oxygen is 32, therefore the mass of oxygen is 8 X 32 = 256 grams
|
Mass-mass stoichiometry
|
|
Methane burns in oxygen to produce carbon
dioxide and water according to the equation above.
Step 2 Calculate the ratio from the prefixes in the balanced equation above
Step 3 Calculate the number of mole of oxygen needed.
Step 4 Calculate the mass of oxygen.
|
|
|