Sulfuric acid has many uses in a modern society. It is used to produce a range of products from fertilisers to car batteries.

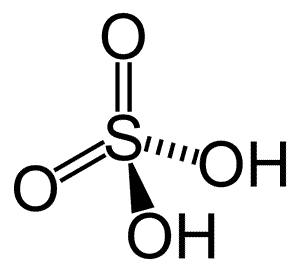
Sulfuric acid is manufactured from molten sulfur or a supply of sulfur dioxide gas in a process called the contact process. A typical manufacturing plant is shown on the right.

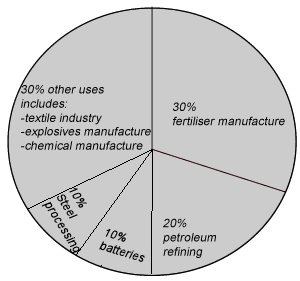
Continue with the uses of sulfuric acid