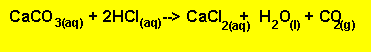
Solution
Step 1 - Work out the equations for all the reactions taking place.
The equation for
the reaction betweenHCl with the limestone is given below.
Equation 1
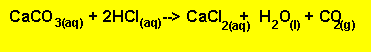
The equation for
the reaction between HCl and sodium carbonate is given below.
Equation 2
![]()
Step 2 - From equation 2 we can work out the mole of HCl left unreacted.
![]()
Step 3 - Work out how much HCl did react. Subtract the amount in excess from the amount originally added.
Amount originally
added - amount in excess = amount reacted.
(0.150 X 0.910) - 0.0504 = 0.0861 mole of HCl reacted.
Step 3 - Using equation
1 calculate the amount of caclium carbonate present.
Mole of HCl /2 = mole of calcium carbonate = 0.0861 / 2 = 0.0430 mole.
Step 4 - Calculate the mass of calcium carbonate present.
0.0430 X formula mass
= 0.0430X 100.0 = 4.30 grams
Step 4 Calculate the percentage
(mass of calcium carbonate
/ mass of sample) X 100 = %
(4.30/5.00) X100 = 86.0%