Gases are made of molecules in constant, high speed, random motion. The molecules collide frequently with the walls of their container, exerting pressure with every collision. Pressure is simply caused by billions of tiny collisions on the wall of the container every second. Pressure is the force applied over a known area. Collisons of molecules with the walls of the container exert a force on the wall surface. The bigger the surface the lower the pressure. Pressure is given by the formula
force/area
If we compress the volume in which the molecules are placed in the pressure
rises. Why? Simply because the molecules have less distance to travel
to collide with the wall so the number of collisions increases. With an
increase in collisions comes an increase in pressure.
Temperature also has an effect on pressure. Have you noticed how the pressure in the tyres of the family car increases on a hot day? As the gas molecules absorb heat they increase in speed and this causes them to collide more often with the walls of the container and exert a greater pressure.
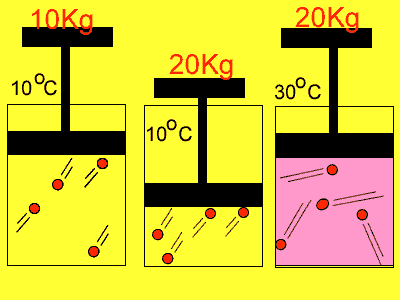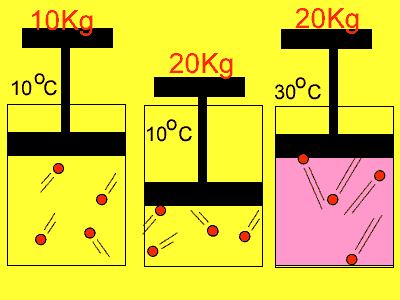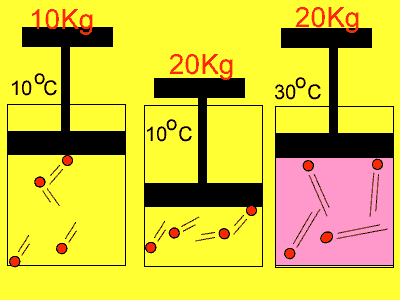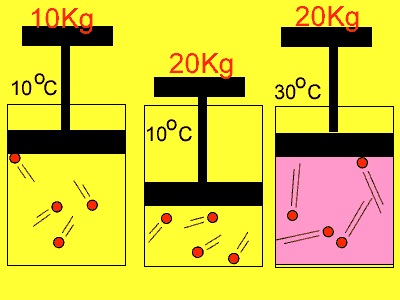
| Reducing the volume of the
gas |
Increasing
the temperature of the gas |
|
| What happens to the speed of the molecules? | |
|
| What happens to the number of collision every second | |
|
| What happens to the pressure of the gas. | |
|