States of matter and hydraulics


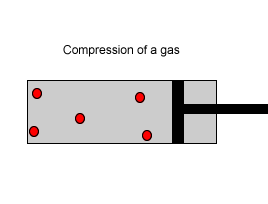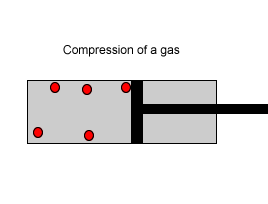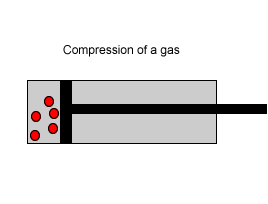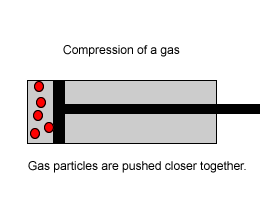
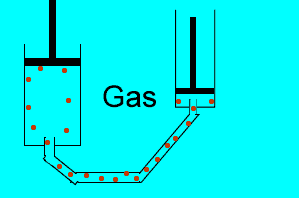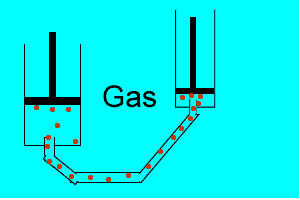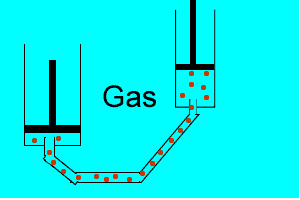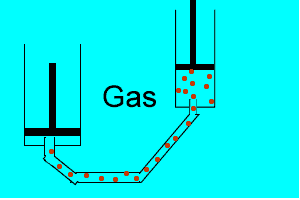
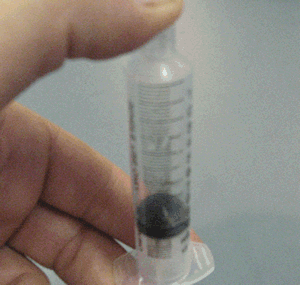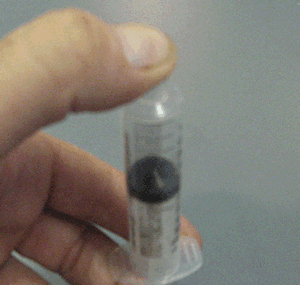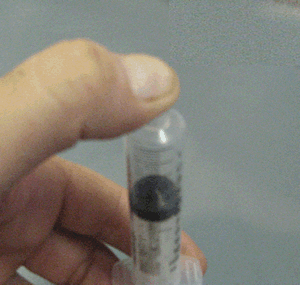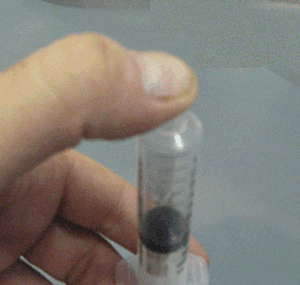
Explain why the plunger of this syringe can be pushed in so far.
Is it filled with a gas or liquid? Explain.
|
States of matter and hydraulics |
|
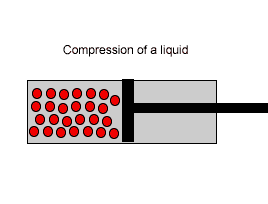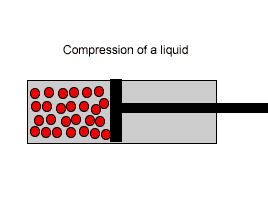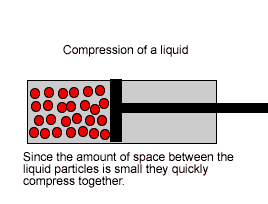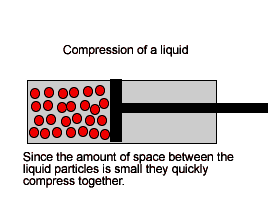
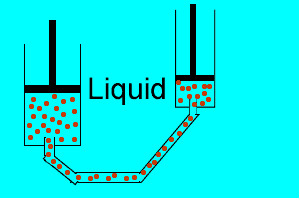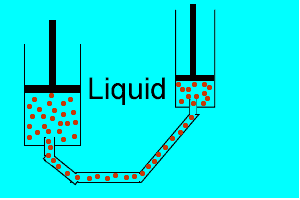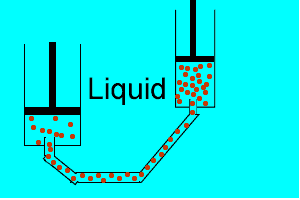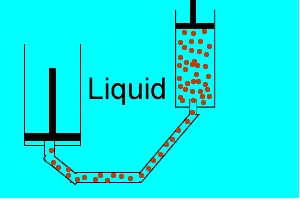
| Liquids are made up of many particles moving randomly amongst each other always maintaining contact. Very little space exists between these particles. |  |
| Gases are composed of high velocity particles that fly in straight paths. The volume of the gas particle is insignificant when compared to the space in which they occupy. |  |
| Consider the animation on the right. Since a gas consists mainly of space, it is very easy to compress. |  |
| Liquids, on the other hand, are composed of molecules where the space between them is not as great as that found in gases. As a result liquids can not be compressed as much as gases. |  |
 |
 |
| Consider thte animation above. As the piston on the left is pushed down the piston on the right responds by moving out. However the piston on the right does not respond as quickly when the tube is full of air as it does when the tube is full of liquid. Why? | |
Explain why the plunger of this syringe can be pushed in so far. Is it filled with a gas or liquid? Explain. |
 |
Home