Science of Conflict
War and the periodic table
This unit was adapted from a book called "The Disappearing Spoon" Chapter 5 "Elements in times of war"
The periodic table is a neat way of representing all the elements on earth such that we can quickly understand the properties of each element.
As you can see elements are arranged in columns, called groups, and rows, called periods.
But what determines in which group or period an element is placed? An atom's electronic structure. Neutral atoms of different elements have a unique number of electrons arranged in a particular way inside the atom. This electronic arrangement determines where on the periodic table the element is placed.
Let's have a look. All elements:in :
group 1 have 1 electron in the top energy level of the neutral atom;
group 2 have 2 electrons in the top energy level of the neutral atom;
.
.
group 7 have 7 electrons in the top energy level of the neutral atom;
group 8 have 8 electrons in the top energy level of the neutral atom.
period 1 have 1 energy level, that contains electrons, in the neutral atom;
period 3 have 2 energy levels, that contain electrons, in the neutral atom;
After placing the elements in the periodic table according to their electronic arrangement we find that elements in the same group have similar properties.
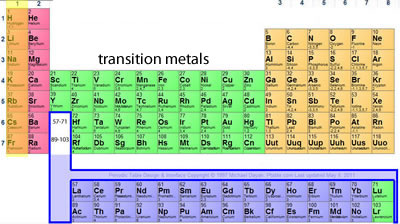
Is bromine more reactive than chlorine?
Which is heavier sodium or potassium?
Which is more reactive sodium or potassium?
Fluorine is found in group 7 period 2 of the periodic table. From this we can tell
Sodium reacts violently with water to produce hydrogen gas. What element would be even more reactive when placed in water?
What element also behaves like carbon to form crystal like structures?
Neon is a very stable element. It hardly reacts with any other element. Which other element is also very stable at room temperature?