Rates (2024 VCE)
These questions can also be found in the 2024 VCE equilibrium topic questions
1) The reaction between iron(III) ions, Fe3+(aq), and thiocyanate ions, SCN- (aq), produces iron(III) thiocyanate ions, FeSCN2+(aq).
![]()
A few drops of sodium thiocyanate solution, NaSCN(aq), are added to 200 ml of the equilibrium mixture at constant temperature.
The concentration-time graph shows the change in concentration of SCN-(aq).
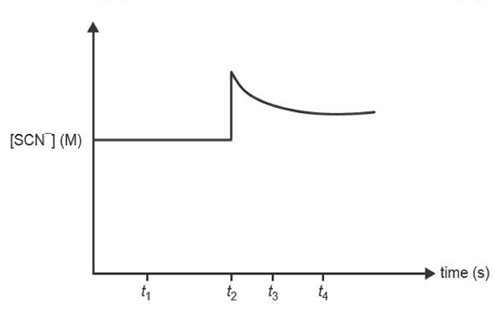
Which one of the following statements is true?
A. The equilibrium constant at t4 is lower than at t1 .
B. The chemical energy of the system increases between t3 and t4 .
C. The rate of the reverse reaction is less than the rate of the forward reaction at t3 .
D. Both the concentrations of Fe3+ and FeSCN2+ are higher at t3 than at t1.
Solution

4) Ammonia gas, NH3(g), can be made by reacting a mixture of nitrogen gas, N2(g), and hydrogen gas, H3(g), using an iron catalyst.
The reaction can be represented by the equation
![]()
The equilibrium constant, KP, at 400 °C is 1.60 x 10-8 kPa-2.
In a mixture that is at equilibrium at 400 °C, the partial pressure of H2 is 88.7 kPa and the partial pressure of N2 is 43.4 kPa.
I
Explain how the iron catalyst addresses the conflict between optimal rate and temperature considerations in the production of NH3 Support your answer with reference to one green chemistry principle. Refer to item 26.ii of the Data Book. 3 marks
Solution
