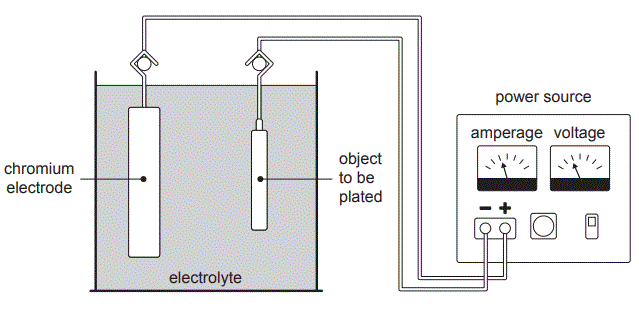
1. Stef is an engineer working in the electroplating industry. Electroplating is a process that uses electrolysis to coat objects with a thin layer of metal to protect the object or improve its appearance. Traditionally objects are electroplated with chromium, Cr, using the set-up shown below.
The electrolyte used is chromium(III) sulfate, Cr2(SO4)3(aq), with a small amount of sulfuric acid, H2SO4(aq).
The half-equation for chromium electroplating is
Cr3+(aq) + 3e- → Cr(s) E0 = −0.74 V
a. Explain, using oxidation numbers, why this is a reduction reaction. (2 marks)
Solution
b. Explain why the effective electroplating of an object requires the same ion to be involved in both the anode and cathode reactions. (2 marks)
Solution
c. Small amounts of H2SO4(aq)) are used in this electroplating cell. Use item 1 of the Data Book to justify why the amount must be carefully managed.
(3 marks)
Solution
d. Over a period of 2.50 hours, a mass of 7.80 g of Cr (Ar = 52.0 amu) was electroplated onto an object. Calculate the current, in amperes, required to achieve this electroplating. (4 marks)
Solution

e. Stef is investigating innovation in the industry and is comparing the use of other metals to chromium in electroplating. Stef has found that rhodium,
Rh (Ar = 102.9 amu), can be used to electroplate parts for wind turbines because it is harder and more resistant to salt corrosion than Cr.
Rh3+(aq) + 3e- → Rh(s) E0 = +0.76 V
Explain why less current would be required to plate the same mass of Rh compared to Cr. (2 marks)
Solution

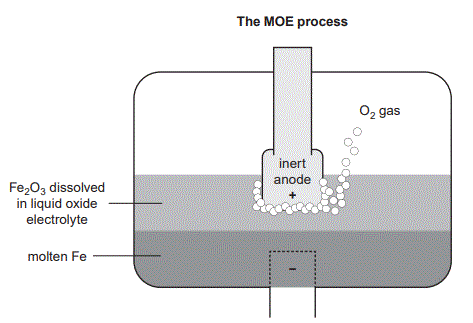
2. Steel is an essential component of modern life. In 2023, approximately 1.9 billion tonnes of steel was made worldwide and steel production was responsible for 8% of global carbon dioxide, CO2, emissions. In traditional steelmaking, iron ore, which is primarily iron(III) oxide, Fe2O3, is reduced with a form of carbon, C, known as coke. This occurs in a blast furnace with temperatures between 2200 °C and 2300 °C, producing molten iron, Fe, and forming CO2 as a by-product. One alternative process being considered is molten oxide electrolysis (MOE). In MOE, Fe2O3 is heated to approximately 1600 °C and dissolved in a liquid oxide electrolyte such as calcium oxide, CaO. Fe is formed at the cathode and oxygen gas, O2(g), is formed at the inert anode. The inert anode needs to be highly stable and corrosion resistant. Materials being considered for the anode include:
• platinum, Pt
• iridium, Ir
• ceramic–metal oxides
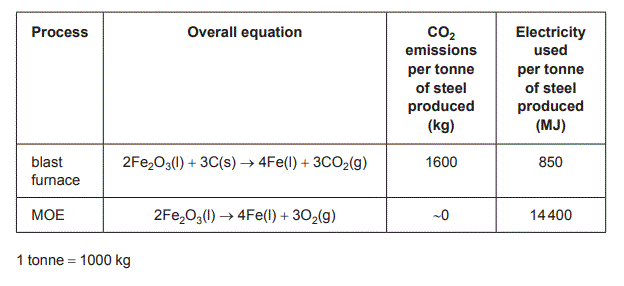
The table below provides a summary of the key characteristics of the two processes.
a. Goal 12 of the United Nations Sustainable Development Goals is ‘responsible consumption and production’. Describe a necessary requirement for the MOE process to have a positive impact on this goal while still meeting society’s needs for steel.
(
2 marks)
b. Evaluate the MOE and the blast furnace processes with respect to green chemistry principles. Refer to item 26 ii of the Data Book, the information on pages 36 and 37 and content you have learnt in VCE Chemistry in your response. Your response must:
• include two relevant green chemistry principles and compare both methods against each principle
• describe one ethical factor that is important to the challenge of producing steel to meet global needs
• include a concluding statement that is consistent with your evaluation. (6 marks)