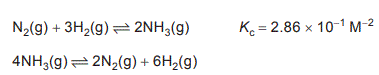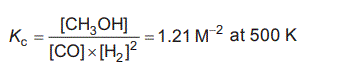| Equilibrium (2025 VCE) | |
1.Consider the following two reactions that are at equilibrium at 500 °C.
The magnitude of the value of Kc for the second reaction is
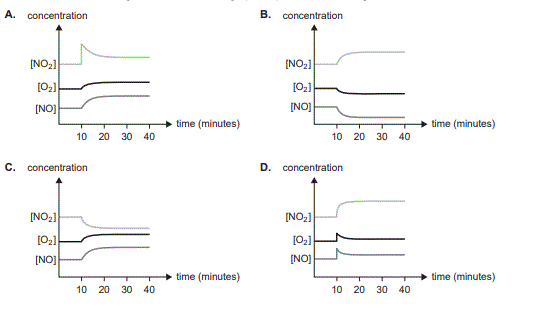
At time t = 10 minutes, the temperature is increased. Which one of the following concentration–time graphs represents the change in concentrations?
Solution |
 |
3. Methanol, CH3OH, is widely used as a starting material for a range of products, including paints and pharmaceuticals. A chemical reaction for the synthesis of CH3OH is
a. Define the term ‘reversible reaction’. (1 mark) i. The rate of the reaction can be increased by increasing the temperature. Explain the impact this would have on the yield. (2 marks) Solution ii. Using Le Châtelier’s principle, state one way in which the yield of
A change to the system is applied at time t1. The concentrations at t1 are measured to be: Determine what needs to happen for the system to return to equilibrium. Use calculations to support your response. (3 marks) d. 5.00 mol of CO and an unknown amount of H2 were injected into an evacuated 1.00 L sealed container at 500 K. Once equilibrium was established, the concentration of CH3OH was determined to be 1.30 M. Calculate the amount of H2, in moles, initially added to the container. Solution |
 |