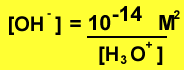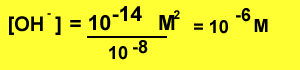pH and Kw
Water will self ionise according to the equation below.
H2O(l) + H2O(l) <=> H3O+(aq) + OH-(aq)
Because of self ionisation water will conduct electricity ever so slightly.
The equilibrium expression for this equation is given below.
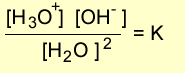
But since the concentration of water molecules in an aqueous solution is virtually constant we can write the expression as
![]()
In pure water,
at 25oC, the [H3O+] = [OH-]
= 10-7
Therefore Kw
= 10-14
The concentration of hydronium and hydroxide ions is very small
and this is why pure water will conduct electricity to a small extent.
This relationship allows us to calculate the concentration of hydronium
and hydroxide ions in solution at 25oC.
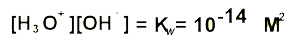
Knowing the hydroxide concentration of a solution we are able to find the pH via the expression above. We can change the expression to solve for the concentration of hydronium ions
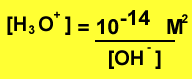
Or we can change the expression to solve for the concentration of hydroxide ions.
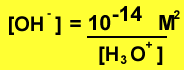
Example. What is the pH of a 0.1 M NaOH solution?

![]()
Example What is the [OH-] of
a solution with pH 8?
Step 1 find the concentration
of hydronium ions
![]()
![]()
Step 2 using the expression
below find the [OH-]