Acid - Base Reactions
An acid base reaction involves the transfer of a proton from the acid to the base. The products are also an acid and a base.
H2SO4(aq) + OH-(aq) <=> HSO4-(aq) + H2(l)
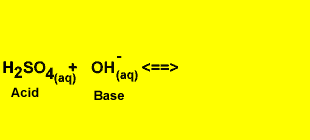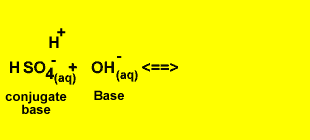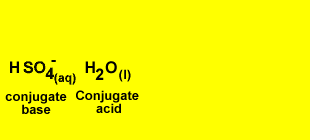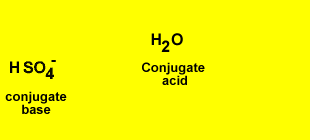
Which of the following reactions are NOT acid/base reactions.
Identify the acid and the base pairs in each acid/base reaction.
Click to see an example
|
Reaction
|
|
|
|
Solution
|
|
Solution
|
|
|
|
|
|
|
|
|
|