Atomic absorption spectroscopy
Instrumentation
Atomic-absorption spectroscopy uses the absorption of light to measure
the concentration of gas-phase atoms. Since samples are usually liquids
or solids, the atoms or ions under investigation must be vaporized in
a flame. The heat of the flame, as well as the action of the reducing gas (fuel), serve to reduce the ions of the sample under investigation to give atoms.
For example Ca2+ + 2e => Ca
The atoms absorb ultraviolet or visible light and their valence electrons make transitions
to higher electronic energy levels. The amount of light absorbed is
directly proportional to the concentration of the atoms under investigation.
Concentration measurements
are usually determined from a working curve after calibrating the instrument
with standards of known concentration.
Exercises
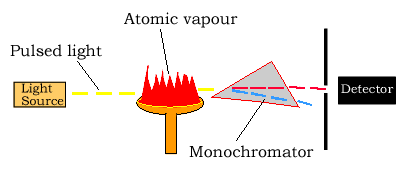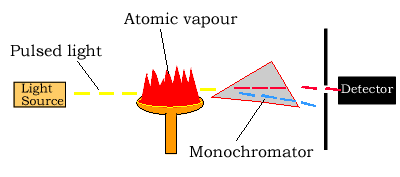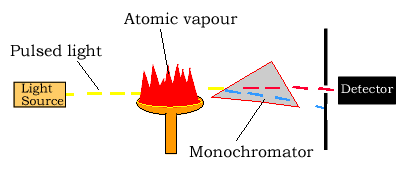
Light source
The light source is usually a hollow-cathode lamp of the element that
is being measured. The disadvantage of these narrow-band light sources
is that only one element is measurable at a time.

Atomizer
Atomic absorption spectroscopy requires that the atoms of the element being investigated be in the gas phase. Ions or atoms in a sample must undergo vaporisation in a high-temperature source such as a flame.
Light separation (Monochromator) and detection
Atomic absorption spectrometers use monochromators and detectors for uv and visible light. The main purpose of the monochromator is to isolate the absorption line from from background light and from molecular emissions originating in the flame.
Sometimes multi-element lamps are used which produce a large number of emitted lines and a monochromator is necessary for isolation of the line of interest.