As we have seen before, atoms will give away and take
electrons from each other. There are two types of atoms, metals and
non-metals. Metals tend to give away electrons to non-metals. A metal
atom needs a non-metal atom to react with. A METAL ATOM WILL NOT REACT
WITH ANOTHER METAL ATOM TO EXCHANGE ELECTRONS. If an atom is to give
up electrons there must be an atom that want to take electrons.
Will a reaction occur between Li and B. - No! Both Li and B want to
give electrons away. There is no atom present to take electrons.
Will a reaction occur between He and F. - No! He is very stable with
two electrons and will not react with any other atom.
Will a reaction occur between Li and F. - Yes! Li will give up an electron
and F will take the electron.
Now, we know that metals and non-metals react together to exchange electrons
but what about two non-metal atoms.
When two non-metal atoms come near each other they will react and share
electrons. Electrons travel so fast over such incredibly short distances
that they can almost be in two places at once.
Take two fluorine atoms for example.
Each atom needs one electron to complete its top level. Neither atom
will give an electron away, so they get close and share an electron
each.
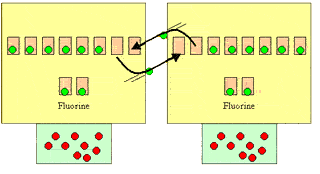
Click to see a more detailed explanation as to why the electrons are shared