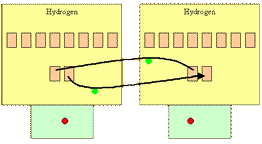
Two hydrogen atoms can come together and react by sharing one electron each. The chemical bond that forms between the two atoms is made up of two electrons, one from each atom.
Exercises
Discuss if a reaction will occur between the following pairs of atoms and explain why or why not. Discuss in terms of give/take or sharing of electrons.
1) Hydrogen and Helium
2) Lithium and Berylium
3) Oxygen and Berylium
4) Fluorine and Neon
5) Fluorine and Oxygen
6) Nitrogen and Fluorine
7) Oxygen and Lithium
8) Berylium and Fluorine
9) A molecule is made up of one oxygen atom and 2 hydrogen atoms. Draw the molecule and explain how the electrons are manipulated between the atoms.
10) A molecule is made up of one carbon atom and 4 hydrogen atoms. Draw the molecule and explain how the electrons are manipulated between the atoms.
11) A molecule has 2 oxygen atoms and 2 hydrogen atoms. Draw the molecule and explain how the electrons are manipulated between the atoms.
12) A molecule has one nitrogen atom, one oxygen atom and one hydrogen atom. Draw the molecule and explain how the electrons are manipulated between the atoms.
13) Berylium is ametal and gives away electrons.
How many electrons will this atom give
away?
The protons have a positive charge while the electrons have a negative
charge. When the berylium atom gives up its electrons what charge does
it form?
Devise a game to reinforce the knowledge gained in this section. The game can be based on a board or card game.