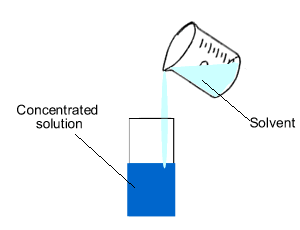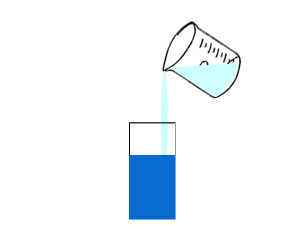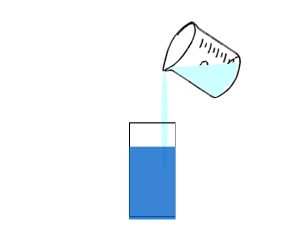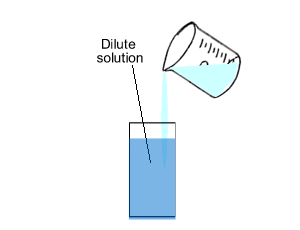
Concentration is the amount of substance, solute, in a given volume of solvent.
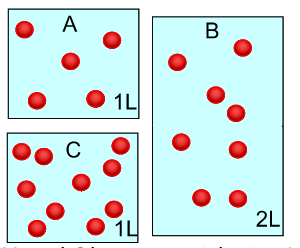
The number of mole in a given
volume of a solution of known concentration is given by the expression
Mole = concentration X volume.
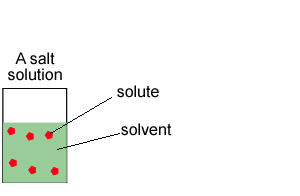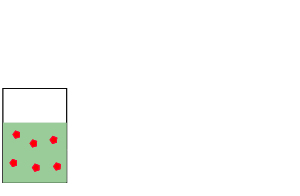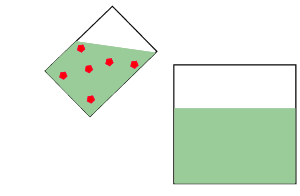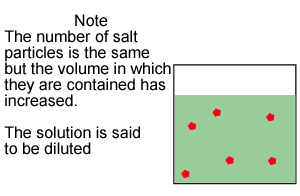
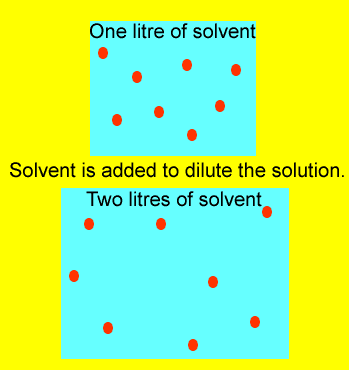
Look at the image on the right.
The number of mole of solute(red balls) remain the same if we dilute (add
solvent) the solution. So
we can generate the expression below.
C1 X V1 = mole = C2 X V2
C1 = initial concentration
V1 = initial volume
C2 = diluted concentration
V2 = larger volume
Lets look at an example.
What volume of water should be added to 200mls of a 0.1M NaOH solution in order to change the concentration to 0.05M NaOH.
What the question is
asking is to find the amount of water that must be added to 200mls of
a 0.1M solution in order to drop the concentration down to 0.05M.
Firstly - Use the formula C1 X V1 = C 2 X V2
Where V1 = 0.2litres ,
C1 = 0.1 and C2 = 0.05
V2 = (0.2 X 0.1)/ 0.05 = 0.4 litres.
Now V2 is the total volume but we wish to find just the amount of solvent added to 200mls. Hence 400-200 = 200mls of solvent must be added to the 200mls of a 0.1M NaOH solution in order to dilute it to 0.05