The
calorimeter
exercise
exercise
.As we found out earlier, in the previous exercise, the calibration factor of our calorimeter is 122.16J/C
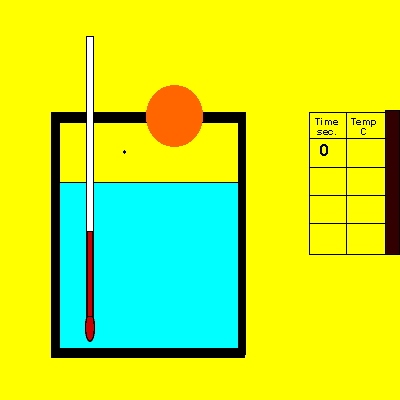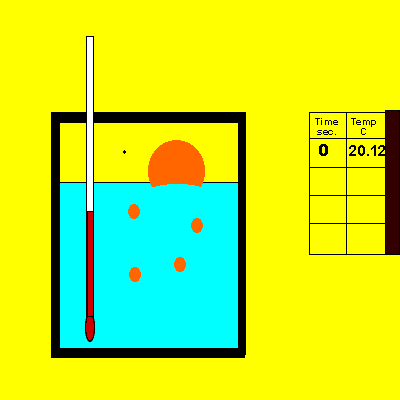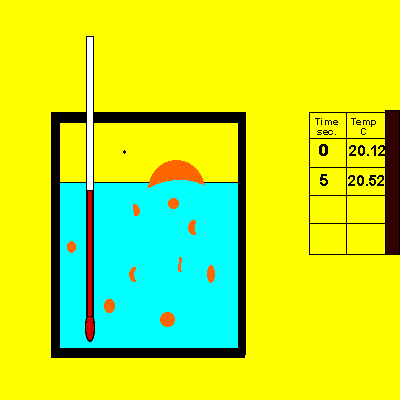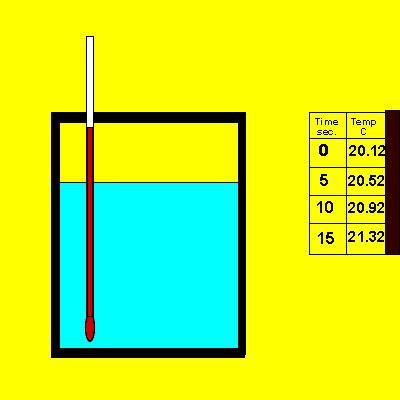
A sample of sodium hydroxide of unknown purity was dissolved in the calorimeter, shown on the right.
a) Is this an exothermic or endothermic reaction? Explain.
b) Calculate the amount of energy that is absorbed or released during this reaction.
Solution