The reaction on the right has reached equilibrium. There is no net formation of product.
What is equilibrium?
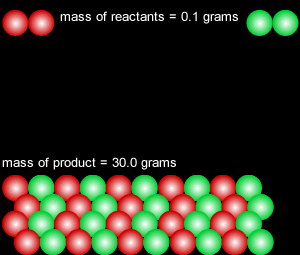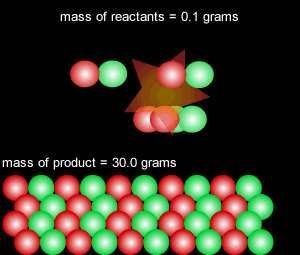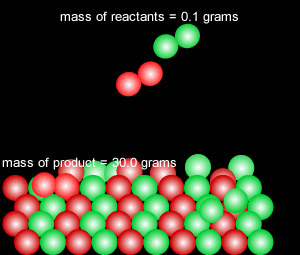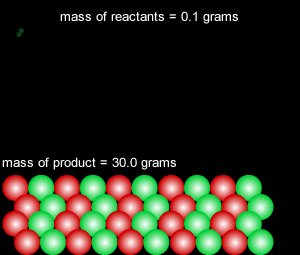
When a reversible chemical reaction takes place in a closed system, in other words a sealed container, it appears that the reaction stops when all the reactants are used up. At the end of the reaction no reactant is left and a maximum amount of product is formed. In a closed system matter is not exchanged with the environment, however, energy is freely exchanged. The image on the right of a terrarium accurately depicts a closed system.
In actual fact the reaction is in a state of dynamic equilibrium. The reverse reaction is occuring at the same rate as the forward reaction and as such the reaction seems to have stopped. No more net product is formed.

sourced from www.youtube.com/watch?v=JsoawKguU6A 14.4.2020