Starch, cellulose and hydrogen bonding
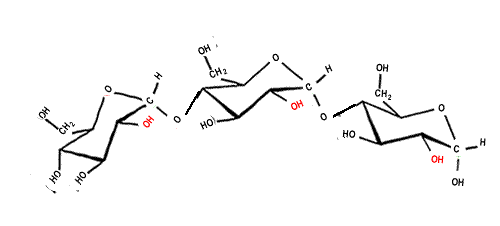
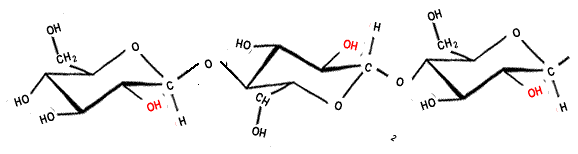
Below is an animation showing where hydrogen bonding occurs along the length of a cellulose chain.
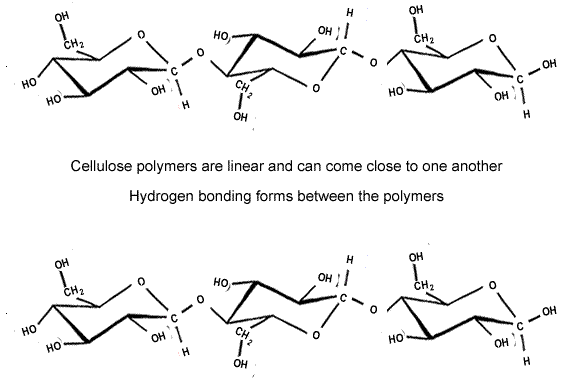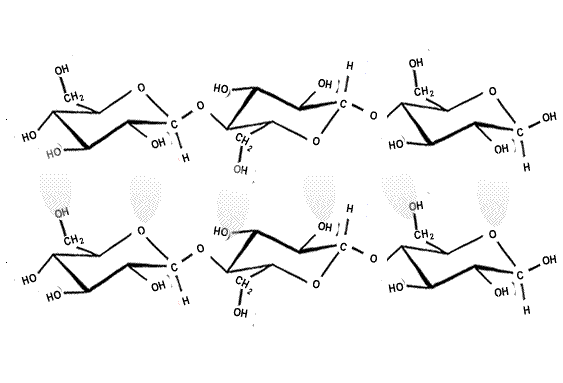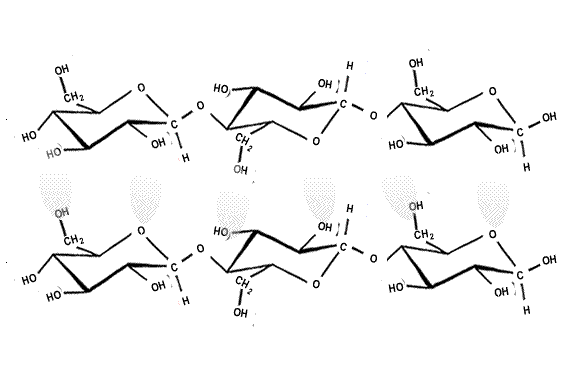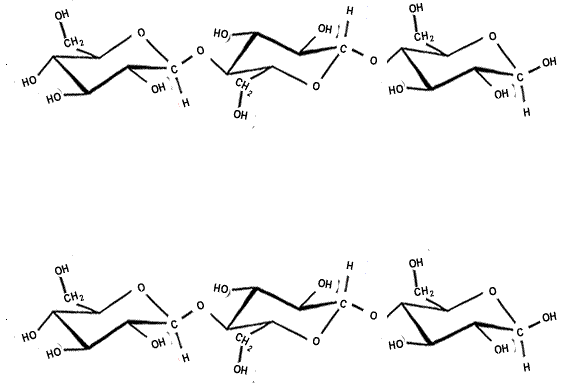
The strength of the covalent bonds withing the polymer chain and the hydrogen bonding between the chains makes cellulose a strong structural material. Up to 2,000 cellulose chains are packed together to form microfibrils, which are packed in overlapping layers, as shown on the right.
The cell wall of a plant cell is composed of cellulose.The polymers are strongly hydrophilic, water loving, which allows the cell wall to become saturated with water, much like a sponge.
