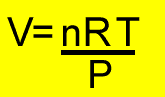A 16.0 gram pure sample of oxygen(O2) was placed in a syringe as shown on the right. The plunger was allowed to expand under atmospheric pressure. The gas was then compressed under a mechanicla force to a pressure of 4 atm. What volume did the gas finally occupy if the temperature of the gas was 17oC?
Atomic
mass of O =16
Clues