Greenhouse Effect
Gases and acid rain.
Global warming may be the long term consequence of using fossil fuels
but the short term effects are felt now. Acid
rain is a direct and immediate consequence of using fossil fuels as
an energy source.

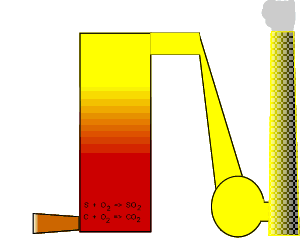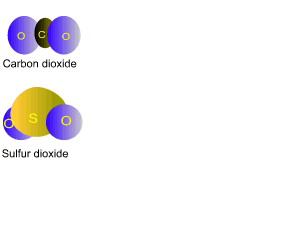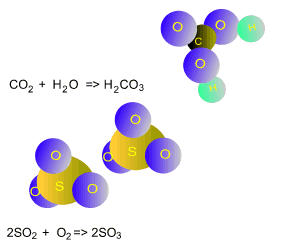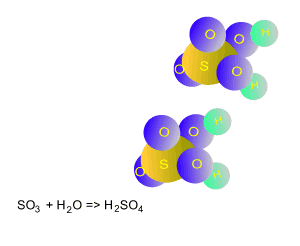
Electricity may look like a clean fuel source but in actual fact it is one of the dirtiest sources of energy.
Coal
is burnt in the power stations and produces carbon dioxide and sulfur
dioxide. Brown coal is a younger fuel then black coal and contains much
less sulfur content. Brown coal from the LaTrobe Valley (Victoria Australia)
has only 0.3%, by mass, sulfur.

Carbon dioxide reacts with water to form carbonic acid according to the equation below. Carbonic acid is a very weak acid.
CO2(g)
+ H2O(l) => H2CO3(aq)
Sulfur dioxide, produced by burning coal, reacts with oxygen and water vapour to form sulfuric acid according to the equations below.
2SO2(g)
+ O2(g) => 2SO3(g)
Sulfur trioxide reacts with atmospheric water to produce sulfuric
acid.
SO3(g) + H2O(l) => H2SO4(aq)