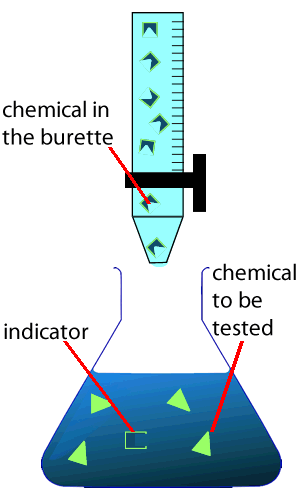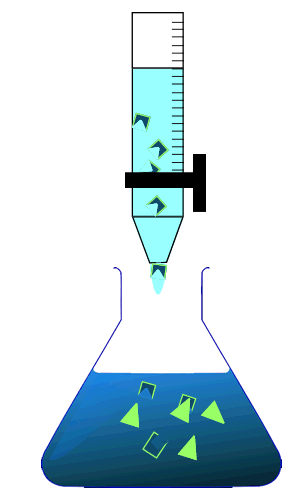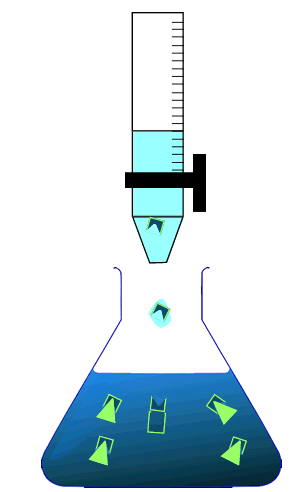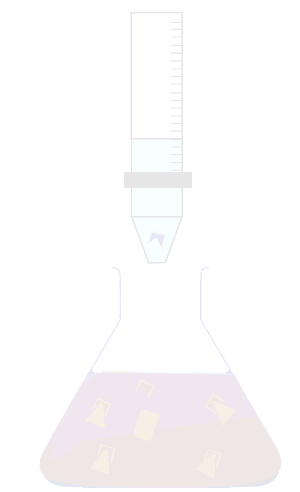
An indicator is a special chemical that changes colour at a certain pH value. It is used when chemists perform a procedure known as a titration to measure accurately the concentration of unknown solutions, for example if a chemist wishes to measure the concentration of acid in a lake.

Scientists use special glassware, as shown on the right, to analyse the concentration of different substances in unknown solutions. The equipment includes:
1) A burette that measures accurately, to 2 decimal places, a variable amount of solution delivered from the burette. This is called the titre.
2) A pipette that measure accurately a certain volume of solution, which may be 10 mL, 20 mL or 25 mL.
3) A conical flask, known as an Erlenmeyer flask.
4) A chemical known as an indicator. The indicator will be explained below in more detail.
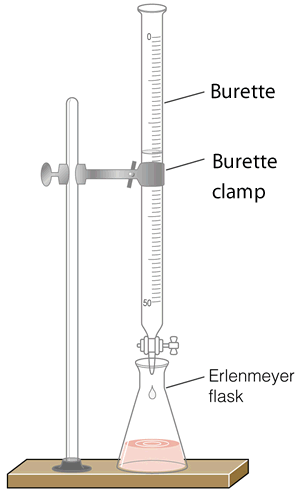
During a titration two solutions, one whose concentration is known accurately and one whose concentration is unknown, are mixed together to try and calculate the concentration of the unknown solution. The solution whose concentration is accurately known is placed in the burette. By measuring the volume of the solution added from the burette we can calculate the concentration of chemical in the unknown solution.
As the reaction taking place is not always visible the trick is when to stop adding the solution from the burette. This is where an indicator is useful.
Consider the animation on the right.
1) An indicator will react with the chemical in the solution placed in the
2) The indicator
3) A colour change occurs when
4) The indicator is always added to the
5) The indicator reacts
6) What is the difference between a 25 mL pipette and burette?
7) When will the indicator change colour?