Nylon
synthesis
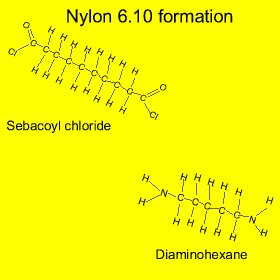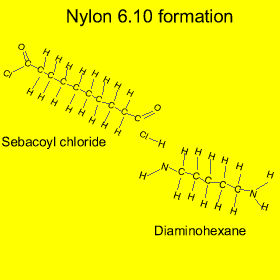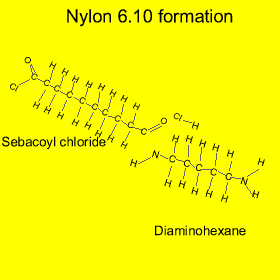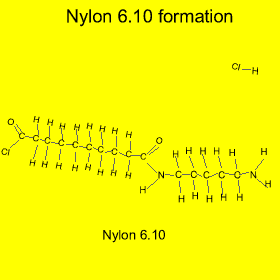
Nylon is formed from a process called "condensation polymerisation" . In this process monomers bond end to end and release a small molecule. In this case hydrochloric acid (HCl) is released, as shown in the animation on the left.
Nylon is also an example of a copolymer as it is formed from two different monomers. Copolymers are polymers formed from two or more different types of monomers.
Fibres formed from nylon are very strong indeed. The rope pictured on the left is made of nylon fibres. Such ropes are extremley strong and used for mountain climbing.

Click to see a 120Kb video of the fibre forming properties.
